Guidelines for the Treatment of Adults with Metastatic Brain Tumors
3. The Role of Whole Brain Radiation Therapy in Adults with Newly Diagnosed Metastatic Brain Tumors
Download PDF Neurosurgery, 2019
Sponsored by: The Congress of Neurological Surgeons and the Section on Tumors
Affirmation of Educational Benefit by: The Congress of Neurological Surgeons and the American Association of Neurological Surgeons
Laurie E. Gaspar MD, MBA1, Roshan S. Prabhu MD, MS2, Alia Hdeib MD3, D. Jay McCracken MD4, George F. Lasker MD, PhD5, Michael W. McDermott MD5, Steven N. Kalkanis MD6, Jeffrey J. Olson MD4
- Department of Radiation Oncology, University of Colorado Denver School of Medicine, Aurora, Colorado, USA
- Southeast Radiation Oncology Group and Levine Cancer Institute, Atrium Health, Charlotte, North Carolina, USA
- Department of Neurosurgery, University Hospitals Cleveland Medical Center, Case Western Reserve University, Cleveland, Ohio, USA
- Department of Neurosurgery, Emory University, Atlanta, Georgia, USA
- Departments of Neurological Surgery, Radiation Oncology, Otolaryngology, University of California San Francisco, San Francisco, California, USA
- Department of Neurosurgery, Henry Ford Health System, Detroit, Michigan, USA
Correspondence:
Laurie E. Gaspar MD, MBA
University of Colorado School of Medicine
Department of Radiation Oncology
Anschutz Cancer Pavilion
Campus Mail Stop: F-706
1665 Aurora Court, Suite 1032
Aurora, CO 80045
Telephone: (720) 848-0115; Fax: (720) 848-0222
E-mail: laurie.gaspar@ucdenver.edu
Disclaimer of Liability
This clinical systematic review and evidence-based guideline was developed by a multidisciplinary physician volunteer task force and serves as an educational tool designed to provide an accurate review of the subject matter covered. These guidelines are disseminated with the understanding that the recommendations by the authors and consultants who have collaborated in their development are not meant to replace the individualized care and treatment advice from a patient's physician(s). If medical advice or assistance is required, the services of a competent physician should be sought. The proposals contained in these guidelines may not be suitable for use in all circumstances. The choice to implement any particular recommendation contained in these guidelines must be made by a managing physician in light of the situation in each particular patient and on the basis of existing resources.
Keywords:
Brain metastases, cerebral metastases, fractionation, histopathology, practice guideline, radiotherapy, surgical resection, whole brain radiation therapy
Abbreviations
BED: Biological equivalent dose
BM: Brain metastases
EGFT: Epidermal growth factor receptor
HVLT: Hopkins Verbal Learning test
Gy: Gray
HA: Hippocampal avoidance
HER2: Human epidermal growth factor receptor 2
MMSE: Mini-mental status examination
NSCLC: Non-small cell lung cancer
PCI: Prophylactic cranial irradiation
QOL: Quality of life
RCT: Randomized controlled trial
RT: Radiation therapy
SCLC: Small cell lung cancer
SIB: Simultaneous integrated boost
SRS: Stereotactic radiosurgery
TKI: Tyrosine kinase inhibitors
WBRT: Whole brain radiation therapy
WHO PS: World Health Organization Performance Status
ABSTRACT
Target population: Adult patients (older than 18 years of age) with newly diagnosed brain metastases.
Question: If whole brain radiation therapy (WBRT) is used, is there an optimal dose/fractionation schedule?
Recommendations:
Level 1: A standard WBRT dose/fractionation schedule (i.e., 30 Gy in 10 fractions or a biological equivalent dose [BED] of 39 Gy10) is recommended as altered dose/fractionation schedules do not result in significant differences in median survival or local control.
Level 3: Due to concerns regarding neurocognitive effects, higher dose per fraction schedules (such as 20 Gy in 5 fractions) are recommended only for patients with poor performance status or short predicted survival.
Level 3: WBRT can be recommended to improve progression-free survival for patients with >4 brain metastases.
Question: What impact does tumor histopathology or molecular status have on the decision to use WBRT, the dose fractionation scheme to be utilized, and its outcomes?
Recommendations: There is insufficient evidence to support the choice of any particular dose/fractionation regimen based on histopathology. Molecular status may have an impact on the decision to delay WBRT in subgroups of patients, but there are not sufficient data to make a more definitive recommendation.
Question: Separate from survival outcomes, what are the neurocognitive consequences of WBRT, and what steps can be taken to minimize them?
Recommendations:
Level 2: Due to neurocognitive toxicity, local therapy (surgery or stereotactice radiosurgery [SRS]) without WBRT is recommended for patients with <4 brain metastases amenable to local therapy in terms of size and location.
Level 2: Given the association of neurocognitive toxicity with increasing total dose and dose per fraction of WBRT, WBRT doses >30 Gy given in 10 fractions, or similar biologically equivalent doses, are not recommended, except in patients with poor performance status or short predicted survival.
Level 2: If prophylactic cranial irradiation (PCI) is given to prevent brain metastases for small cell lung cancer, the recommended WBRT dose/fractionation regimen is 25 Gy in 10 fractions, and because this can be associated with neurocognitive decline, patients should be told of this risk at the same time they are counseled about the possible survival benefits.
Level 3: Patients having WBRT (given for either existing brain metastases or as PCI) should be offered 6 months of memantine to potentially delay, lessen, or prevent the associated neurocognitive toxicity.
Question: Does the addition of WBRT after surgical resection or radiosurgery improve progression-free or overall survival outcomes when compared with surgical resection or radiosurgery alone?
Recommendations:
Level 2: WBRT is not recommended in WHO performance status 0-2 patients with up to 4 brain metastases because, compared with surgical resection or radiosurgery alone, the addition of WBRT improves intracranial progression-free survival but not overall survival.
Level 2: In WHO performance status 0-2 patients with up to 4 brain metastases where the goal is minimizing neurocognitive toxicity, as opposed to maximizing progression-free survival and overall survival, local therapy (surgery or radiosurgery) without WBRT is recommended.
Level 3: Compared with surgical resection or radiosurgery alone, the addition of WBRT is not recommended for patients with more than 4 brain metastases unless the metastases’ volume exceeds 7 cc, or there are >15 metastases, or the size or location of the metastases are not amenable to surgical resection or radiosurgery.
INTRODUCTION
Rationale
Whole brain radiation therapy (WBRT) has long been a standard treatment for patients with brain metastases. Based on preclinical and observational data, some physicians alter dose fractionation or withhold WBRT, based on tumor histology. Concern has also been expressed by clinicians regarding the neurocognitive effects of WBRT, particularly if the metastases are amenable to surgical resection or stereotactic radiosurgery (SRS).
This guideline is based on a systematic review of the evidence available for WBRT dose fractionation regimens and the impact of tumor histopathology on treatment outcomes when WBRT is used for newly diagnosed brain metastases. Due to concerns about neurocognitive toxicity from WBRT, this guideline also reviews the evidence for pharmacologic or technical maneuvers to reduce this toxicity. In addition, this guideline analyzes the data regarding survival outcomes following local therapy with surgical resection or SRS.
Objectives
This guideline will systematically review the evidence available for altered WBRT dose fractionation and the impact of tumor histopathology on treatment outcomes when WBRT is used. The neurocognitive effects of WBRT, and the strategies for reducing these effects, are addressed. In addition, this guideline will also systematically review the evidence for the use of surgical resection plus WBRT compared with WBRT alone in patients with newly diagnosed, surgically accessible, single brain metastases. The studies identified through this process will be used to make evidence-based recommendations for the role of WBRT in the management of patients with newly diagnosed brain metastases.
METHODS
Writing Group and Question Establishment
The writing group was established by the nominating section and Task Force Chair. The writing group jointly developed the 4 questions relevant to WBRT in the current era. The 4 questions were each assigned to a primary writer. To answer the questions, a comprehensive systematic literature review was performed. Two writers evaluated citations found by the search using a priori criteria for relevance and documented decisions in standardized forms. Cases of disagreement were resolved by a third reviewer. The same methodology was used for full-text screening of potentially relevant papers. Studies that met the eligibility criteria were data extracted by one reviewer and the extracted information was checked by a second reviewer.
Literature Review
To update questions raised in the prior guidelines, PubMed, Embase, and Cochrane CENTRAL databases were searched for the period from January 1, 2008, to December 31, 2015. For the new question regarding neurocognitive effects, the search extended between January 1, 1990, through December 31, 2015. A broad search strategy using a combination of controlled vocabulary and text words was employed. The search strategies for each database are documented in Table 1.
Article Inclusion and Exclusion Criteria
For new literature to be included for consideration, studies published in full as peer review papers had to meet the following criteria:
- Be published in English with a publication date within the periods described above.
- Involve patients with newly diagnosed parenchymal brain metastases.
- Involve adult patients (>18 years of age).
- Fully-published peer-reviewed articles.
- Use of WBRT after diagnosis of brain metastases has been made.
Study Selection and Quality Assessment
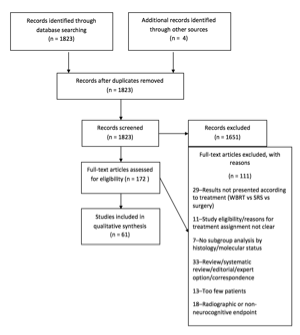
After an extensive search, 1823 articles were found. The duplicates from the searches in different databases were eliminated. By reviewing the titles and/or abstracts, we excluded all articles referring to leptomeningeal metastases, those discussing exclusively surgery, chemotherapy or radiosurgery and citations that only referred to patients <18 years of age. We also excluded publications that discussed exclusively WBRT for treatment of recurrent/progressive brain metastases, and all articles discussing experimental therapy in animal tumor models. The remaining 172 articles underwent full-text review. Only 61 articles met all of the inclusion criteria and were considered in formulating these evidence-based clinical guidelines. The remaining 111 articles that underwent full-text review were excluded for the following reasons: the results were not presented according to treatment type, the study eligibility or reasons for treatment assignment were not clear, a lack of subgroup analysis by histology or molecular status, the paper was a review, systematic review, letter, or editorial, the study contained too few patients, or the study included a radiographic or non-neurocognitive endpoint.
Evidence Classification and Recommendation Levels
Both the quality of the evidence and the eventual strength of the recommendations generated by this evidence were graded according to a 3-tiered system for assessing studies addressing diagnostic testing as approved by the American Association of Neurological Surgeons (AANS)/
Congress of Neurological Surgeons (CNS) Joint Guidelines Review Committee on criteria
Assessment for Risk of Bias
A list of article titles and abstracts was produced by the search, using the search strategies presented in Table 1. To avoid bias due to selective choice of articles, the decision to review and utilize the full article was made by at least 2 authors. The authors of this guideline represent multiple specialties. The decision to classify a study as Class I, II, or III was first made by the primary author of each of the 4 questions, and then reviewed by at least 1 other author. The strength of the recommendation was also proposed by the primary author and then discussed and modified by all authors.
RESULTS
If WBRT is used, is there an optimal dose/fractionation schedule?
In the 2010 guideline, 17 studies met the eligibility criteria for this question.1 These unique studies fell into 3 evidence class categories as follows: 9 randomized controlled trials (RCT) Class I studies 2-10 and 1 Class II randomized phase I/II trial, 11 7 other Class II studies 12-17 (retrospective cohort studies), and 1 Class III study 18 (prospective cohort study with historical controls). Since 2008, there have been 3 additional studies that met eligibility criteria: 1 Class I study 19 and 2 Class III studies. 20 , 21 Table 2 summarizes the 14 RCT studies from the old and new guidelines that informed the recommendations.
Expressing radiation dosages in terms of the biological equivalent dose (BED) takes into account the total dose of radiation, fraction size, and overall time to deliver the radiation, and presumed repair of irradiated tissue. 22 , 23 The 2010 guidelines found no meaningful improvement in any endpoint relative to dose or BED; specifically, survival was not improved. In addition, no dose-effect was identified for quality of life (QOL) or neurologic function. Given the paucity of Class I studies published since the 2010 guidelines, these BED analyses were not updated.
Despite previously published phase III studies (all Class I studies) finding no disadvantage to very short, accelerated treatments, there have been few recent studies evaluating this further. 2 , 6, 10 One recent phase II study of short accelerated radiation therapy (RT), such as 18 Gy given in 4.5 Gy fractions twice daily for 2 days, concluded that this treatment was effective in terms of symptom relief (63%) and median survival time (7 months), but agreed that further phase III studies were required.24
One of the evolving techniques of WBRT is to use a simultaneous integrated boost (SIB). 25 , 26 The decision to do a SIB may be based on the size of the brain metastases or histology of the primary cancer. 27 Rodrigues et al 25 reported on such a technique for 120 patients with oligometastatic brain metastases (< 7 lesions with cumulative volume < 30 cc) treated at 2 centers between 2005 and 2010. Using an arc-based image-guided system, patients received 20 Gy in 5 fractions WBRT while simultaneously receiving 40 Gy in 5 fractions to the oligometastases. With a median follow-up of 4.7 months, 23% of deceased patients died of intracranial disease. The median survival time was 5.9 months. As in other WBRT studies, poor performance status, lung cancer histology, and the presence of systemic disease were identified as poor prognostic factors. A phase II study comparing this technique to traditional SRS techniques is ongoing in Canada ( NCT01543542).
In summary, a standard WBRT dose/fractionation schedule (i.e., 30 Gy in 10 fractions or a BED of 39 Gy10) is recommended because altered dose/fractionation schedules do not result in significant differences in median survival or local control. However, due to concerns regarding neurocognitive effects, higher dose per fraction schedules (such as 20 Gy in 5 fractions) are recommended only for patients with poor performance status or short predicted survival. The more difficult issue is when to recommend WBRT. As seen throughout the following questions, the role of WBRT has declined, because more patients are treated with local therapies (radiosurgery or surgery) or supportive care. Studies of local therapy with or without WBRT have only been conducted in patients with <4 brain metastases.28 This lead to the Level 3 recommendation of WBRT to reduce progression-free survival for patients with >4 brain metastases. The use of systemic therapy only is addressed more thoroughly in other chapters.
What impact does tumor histopathology or molecular status have on the decision to use WBRT, the dose fractionation scheme to be utilized, and its outcomes?
In the 2010 guidelines, only 1 paper met the eligibility criteria for this slightly modified question. 29 The question was reworded in this guideline to address the issue of timing of WBRT relative to systemic therapy. This updated literature search identified 3 additional papers, all Class II or III. 30-32 In addition, an older Radiation Therapy Oncology Group (RTOG) Class I study primarily asking a question regarding dose/fractionation was considered because it stratified patients according to site of primary cancer (lung vs breast vs other). 33
Borgelt et al, 33 in a Class II study, concluded that the results of WBRT were no different between 3 histopathology groups: lung, breast, or “other.” No regimen was shown to be superior over another regimen according to these histopathology groups. However, a later retrospective analysis of RTOG and multi-institutional data has uncovered diagnosis (histology) specific prognostic factors. 34 This retrospective analysis of 3940 patients with newly diagnosed brain metastases led to the Graded Prognostic Assessment Index that can be used to estimate survival for patients with brain metastases from non-small cell lung cancer (NSCLC), small cell lung cancer (SCLC), melanoma, renal cell cancer, breast cancer, or gastrointestinal cancers. Because these patients had undergone a variety of treatments, including WBRT, SRS, surgery, and various combinations, the authors were careful to conclude that although histology may influence prognosis, there were insufficient data to predict the relative benefits of one treatment over another.
Lung cancer has been identified in several studies to have a different outcome when treated with WBRT than other histologies. In RTOG 9508, patients with 1 to 3 newly diagnosed brain metastases were randomized to receive either WBRT or WBRT followed by a SRS boost. 35 The primary study outcome was overall survival, and secondary outcomes were tumor response, local control rates, overall intracranial recurrence rates, cause of death, and performance measurements. No difference between WBRT alone versus WBRT followed by SRS was found in these primary or secondary endpoints for the study group at large. However, a subset analysis found improved survival, which reached statistical significance in multivariate analysis, for patients who received the combination of WBRT and SRS, as opposed to WBRT alone, in squamous cell and non-small-cell histology, which is usually seen in patients with lung cancer.
The molecular analysis of lung cancer has also brought about significant changes in the approach to brain metastases with either epidermal growth factor receptor (EGFR) mutations or echinoderm microtubule-associated protein-like 4/anaplastic lymphoma kinase (ALK) rearrangements. 36 , 37 Two small institutional retrospective Class III case series of patients with lung cancer brain metastases treated with WBRT attempted to determine the impact of EGFR mutation in treatment outcome. 30 , 31 Both studies found that an EGFR mutation was predictive for improved treatment response following WBRT. Gow et al 31 also concluded from a small retrospective study that the addition of a tyrosine-kinase inhibitor to WBRT was independently associated with improved treatment response in EGFR-mutated patients. Small retrospective studies in EGFR-mutated lung cancer patients have found that first-line tyrosine kinase inhibitors (TKI) without WBRT are associated with response or stability in brain metastases, but that intracranial progression requiring WBRT occurs in most patients. 38 Despite the controversy regarding treatment for this subset of lung cancer patients, there are no ongoing phase III studies comparing WBRT to TKIs in EGFR-mutated or echinoderm microtubule-associated protein-like 4/anaplastic lymphoma kinase rearranged patients.
Molecular analyses in patients with breast cancer have also uncovered the importance of human epidermal growth factor receptor 2 (HER2) status on the outcome of patients with breast cancer brain metastases undergoing WBRT. In a class III study, Wolstenholme et al 32 reported the results of WBRT observed in 88 HER2-positive patients and 93 HER2-negative patients, with heterogeneous chemotherapy regimens, including trastuzumab treatment in 53 of the 88 HER2-positive patients. Twelve patients also received additional SRS. The study concluded that an improved median survival following WBRT was associated with HER2-positive status. However, the results were confounded by the observation that HER2-positive patients may have had more aggressive treatment for their brain metastases.
Though this systematic review of the literature was limited in terms of higher class data that specifically addressed the question of the impact of histopathology/molecular status on treatment outcomes following WBRT, it appears that the use of WBRT has waned, particularly in certain primary histologies. For example, several retrospective Class III case series have concluded that SRS alone for melanoma brain metastases, even if numerous, is associated with a reasonable outcome. 39-41 Prospective studies are needed, and a randomized prospective trial investigating the role of WBRT in melanoma brain metastases is reported to be underway. 42
In summary, there is insufficient evidence to support the choice of any particular dose/fractionation regimen based on histopathology. Molecular status may have an impact on the decision to delay WBRT in subgroups of patients but there are not sufficient data to make a more definitive recommendation. The role of WBRT, as opposed to SRS alone, is also controversial in many histologies, but particularly for patients with melanoma. RCTs that are histology- or molecular status-specific are necessary to resolve many of these issues.
What are the neurocognitive consequences of WBRT, and what steps can be taken to minimize it?
This is a new question since the prior guidelines were published, reflecting the growing concern about the neurocognitive effects of WBRT. The effects of WBRT on neurocognitive functions can be subdivided into whether or not patients have demonstrable brain metastases at the time of WBRT, or whether WBRT is being used for prophylactic cranial irradiation (PCI). Six studies of the neurocognitive effects of WBRT in the PCI setting for SCLC are summarized in Table 4. 43-48 These studies primarily included patients with SCLC histology, although Sun et al 45 reported on the neurocognitive outcome of PCI in patients with NSCLC.
An early phase III trial by Arriagada et al. 48 reported neurocognition as a secondary endpoint for patients with limited stage SCLC. There was no difference found in the 2-year cumulative incidence of negative change in cognitive “higher functions” (36% if no PCI, vs 30% with PCI, p = NS). This study was given a Class II designation due to the lack of definition for “higher functions” testing, or criteria used to define decline in testing. Gregor et al. 47 also found no difference in neurocognition at 6 months or 1 year following PCI. This RCT was given a Class II designation for several reasons: neurocognition was only a secondary endpoint, and neurocognitive baseline testing was available in only 40% of patients, leading to potential issues of selection bias and small patient numbers. Slotman et al. 46 reported neurocognition within a phase III RCT for patients with extensive stage SCLC. There was no statistical difference in worsened cognitive functioning at 3 months (PCI: 22.4% versus no PCI: 10%, p = NS). This study had a large number of patients treated with a PCI dose/fractionation scheme not as frequently used in the United States (20 Gy in 5 fractions). Another limitation was that the neurocognitive endpoint was taken from a subset of primarily QOL questionnaires. Sun et al. 45 reported the neurocognitive outcomes in an RCT of PCI or no PCI for NSCLC histology. Patients in the PCI arm had a significant deterioration in memory, measured by the Hopkins Verbal Learning Test-Revised (HVLT-R), at 1 year. However, there was no difference found in global cognition measured by the Mini-Mental Status Examination (MMSE) or QOL between arms. This study represents Class I data due to a relatively large patient population, intact randomization, and the use of more sensitive neurocognitive testing.
Two studies investigated the cognitive effect of various PCI dose/fractionation regimens for patients with PCI. 43 , 44 Le Pechoux et al 44 found no significant difference in neurocognitive outcomes between 36 Gy and 25 Gy PCI. However, Wolfson et al 43 reported secondary endpoints of a large randomized phase II trial using a modern battery of neurocognitive assessments and reported a significantly higher rate of neurocognitive decline with 36 Gy versus 25 Gy at 12-months (85 – 89% vs 60%, p = 0.02). Increasing age was also a significant predictive factor for neurocognitive decline. Thus, the class II evidence from the Wolfson et al 43 study allows one to infer that WBRT doses exceeding 30 Gy in 10 fractions (or similar BEDs) are associated with greater likelihood of neurological decline.
Three studies summarized in Table 5 met inclusion criteria for tracking neurocognitive outcome following local brain therapy (primarily SRS) versus local brain therapy and WBRT for patients with known brain metastases. 49-51 Chang et al50 randomized patients with 1 to 3 brain metastases to SRS alone versus SRS and WBRT. A sensitive battery of neurocognitive assessments was utilized with neurocognition as the study’s primary endpoint. The study showed significantly higher rates of deterioration in recall at 4 months with the addition of WBRT (SRS + WBRT: 52% vs SRS: 24%, p(A > B) 96%). Another study by Aoyama et al 49 randomized patients with 1 to 4 brain metastases to SRS versus SRS and WBRT, and used the MMSE as a measure of global cognition. This study found no difference in MMSE preservation rates between arms at both 12 and 24 months. In fact, they showed that intracranial tumor control was the most important factor in cognitive preservation. In a more recent study, Brown et al 52 similarly showed that the addition of WBRT to SRS was associated with significantly higher rates of cognitive decline and memory decline at 3 months (SRS + WBRT 92% vs SRS 64%, p<0.001).
Soffietti et al 51 reported the secondary cognitive outcome of local therapy (SRS or surgery) with or without WBRT in an RCT by the European Organisation for Research and Treatment of Cancer (EORTC). The authors reported that WBRT was associated with significantly more decline in 12-month cognitive functioning than local therapy alone. This trial was graded as Class II due to the use of primarily QOL questionnaires to measure cognition and the mixing of post-surgical and SRS local therapy patients into a single group.
Four studies summarized in Table 6 met the inclusion criteria for medications or radiation techniques evaluated for their efficacy in minimizing the neurocognitive effects of WBRT for patients with known brain metastases.53-56 Three of these trials investigated the use of medications to mitigate the neurocognitive effects of RT in patients with known brain metastases or primary brain tumors. 54 , 55 , 56 Butler et al55 reported an RCT of methylphenidate versus placebo, with approximately 50% of patients having metastatic brain tumors. MMSE was used as the primary measure of cognition. There were no differences in MMSE scores between arms<8 weeks post-radiation. Brown et al 56 reported a phase III RCT of memantine versus placebo in patients with brain metastases treated with WBRT. There was no significant difference in the decline of delayed recall (the primary endpoint) in the memantine arm compared with the placebo arm. However, time to cognitive failure, defined as the first cognitive failure on any of the neurocognitive tests, was found to significantly favor the memantine arm (hazard ratio, 0.78, p=0.01). Rapp et al53 reported a phase III trial of donepezil versus placebo for patients with metastatic or primary brain tumors status post-completion of partial brain RT or WBRT. Patients in both groups showed improved cognitive function at 24 weeks, but there was no significant difference in overall cognitive composite score between the donepezil and placebo arms (p=0.48). However, several specific cognitive functions, such as immediate and delayed recall, did show improvement, and patients with greater baseline impairment were more likely to have the greatest benefit from donepezil.
Gondi et al54 reported a single arm phase II trial of hippocampal avoidance WBRT (HA-WBRT). The results of this trial were compared with a historical control of conventional WBRT. HA-WBRT was associated with a lower rate of decline in delayed recall at 4 months, 7% with HA-WBRT as opposed to 30% in historical control, p=0.0003.
In summary, there is evidence that the addition of WBRT to local therapy (primarily SRS) is associated with increased risk of significant neurocognitive decline in patients with brain metastases. This decline is apparent as early as 3 months post-WBRT and can persist in long-term survivors. This supports a Level 2 recommendation that local therapy (surgery or SRS) without additional WBRT is recommended for patients with <4 brain metastases that are amenable to local therapy in terms of size and location. The evidence also supports a Level 2 recommendation that WBRT doses not exceed 30 Gy given in 10 fractions, or similar BEDs except in patients with poor performance status or short predicted survival. WBRT given as PCI also has detrimental effects on neurocognition, although these detrimental effects have to be weighed against the small survival benefit of PCI. 57 There is evidence that higher doses of PCI are associated with higher levels of neurocognitive detriment, particularly in older patients. 43 , 44 This supports the Level 2 recommendation that the recommended PCI WBRT dose/fractionation regimen is 25 Gy in 10 fractions, and because this can be associated with neurocognitive decline, patients should be told of this risk at the same time they are counseled about the possible survival benefits.
There is Class I evidence that memantine has a nonsignificant trend towards neurocognitive protection in patients with brain metastases undergoing WBRT. This supports the Level 3 recommendation to place patients having WBRT (given for either existing brain metastases or as PCI) on 6 months of memantine to potentially delay, lessen, or prevent the associated neurocognitive toxicity. The evidence for donepezil is moderate, and there is insufficient evidence that methylphenidate is beneficial. There is additional evidence suggesting that HA WBRT may significantly reduce the risk of neurocognitive decline compared with conventional WBRT. There are ongoing RCTs of WBRT with or without HA for patients with either known brain metastases or receiving WBRT in the PCI setting.
Does the addition of WBRT after surgical resection or radiosurgery improve progression-free or overall survival outcomes when compared with surgical resection or radiosurgery alone?
This is a new question raised since the publication of the 2010 guidelines in which there was insufficient evidence to address the value of WBRT following SRS.1 The previous guidelines only addressed surgical resection and WBRT, or WBRT alone. In this guideline, the authors have expanded the scope of treatment and have the results of studies of local therapy, including either surgery or SRS, with or without WBRT. Prospective RCTs addressing this issue are summarized in Table 7. 28 , 58, 59 Sahgal et al60 published a 2015 meta-analysis evaluating SRS and WBRT compared with SRS alone. While this study was not included in our data table as primary evidence, conclusions gleaned from this study are relevant to this review. Since an earlier question addressed the neurocognitive outcomes of WBRT, this question addresses progression-free or overall survival outcomes.
The first large-scale, prospective RCT demonstrating the efficacy of WBRT following neurosurgical resection of a single solitary BM was reported by Patchell et al 58 in 1998. The primary endpoint was intracranial disease control. Improved local control and cumulative intracranial control were observed in patients who received postoperative WBRT when compared with patients who did not receive the adjuvant therapy. Local tumor recurrence in the resection cavity, as well as distant intracranial metastatic disease, was reduced in the patients who received WBRT, as opposed to those who did not. There was also a significant decrease in the incidence of death resulting from neurological sequelae in patients who received WBRT. Although there was no significant difference found between the adjuvant WBRT versus observation groups in terms of overall survival or length of functional independence, the primary endpoint measured in this study was metastatic recurrence in the brain, and the sample sizes were likely underpowered for these analyses.
An RCT published in 2006 by Aoyama et al 59 (JROSG99-1) randomized 132 patients with 1 to 4 brain metastases, each <3 cm in diameter, to receive either SRS alone or SRS and WBRT. The primary endpoint was overall survival, but secondary outcomes included local recurrence, rate of salvage brain treatment, functional preservation, toxic effects, and cause of death. In the SRS only group, median survival time and the 1-year actuarial survival rate were not significantly different from the SRS and WBRT group. Intracranial recurrence rate at 1 year was higher in the SRS group than the SRS and WBRT group (76.4% vs 46.8%, p<0.001). Salvage brain treatment was significantly higher in the SRS alone group; however, the incidence of neurologic-related deaths was not statistically significant. The authors concluded that the addition of WBRT to SRS therapy improved local and intracranial control but did not improve overall survival.
The EORTC 22952-26001 trial, as described by Kocher et al 28 in 2011, randomized 359 patients, WHO performance status of 0-2, who had received local therapy (either SRS or surgical resection of <3 brain metastases) to either the local therapy only or local therapy followed by WBRT. The primary endpoint was time to decline to WHO Performance Status (WHO PS) > 2. Secondary endpoints included frequency and location of intracranial relapse, progression-free survival, and overall survival. The investigators reported that within the surgical subgroup, adjuvant WBRT reduced the probability of both local and distal relapse to new intracranial sites when compared to patients who did not receive WBRT (59% to 27%, p<0.001 and 42% to 23%, p=0.008, respectively). In the pooled analyses of surgery and SRS, the median time to WHO PS > 2 was 10.0 months in the local therapy only arm and 9.5 months in the local therapy and WBRT arm (p=0.71). In a multivariate analysis, the only factors significantly impacting WHO PS outcomes were the baseline WHO PS (0 vs 2, p=0.004) and the presence of macroscopic tumor outside the brain (absent vs present, p<0.001). Median progression-free survival was not significantly longer in the WBRT arm when compared with the observation arm (4.6 months vs 3.9 months, p=0.20). Overall survival was similar between the two arms. Death resulting from neurologic sequelae was significantly greater in the local therapy arm. Systemic disease progression was the most common cause of death in both arms of the study. The results from this RCT provide further evidence that WBRT is an effective modality to decrease intracranial metastatic recurrence and neurologic death, but this does not translate to an improved duration of functional independence or overall survival. The investigators concluded that in well-performing patients with stable systemic disease and <3 brain metastases, WBRT could be withheld if serial imaging is performed.
The North Central Cancer Treatment Group Alliance N0574 Trial was reported by Brown et al 52 in 2016, falling outside the reference search window, and therefore was not utilized when forming the recommendations. 52 This prospective, multi-institutional RCT was designed to investigate the effect of adjuvant WBRT on cognitive function in patients with 1 to 3 BM treated with SRS. This study was graded as Class II evidence because secondary endpoints included time to intracranial failure, QOL, treatment toxicity, functional independence, individual cognitive assessment outcomes, long-term cognitive status, and overall survival. It was shown that patients who received adjuvant therapy experienced significant deterioration in cognitive function and quality of life at 3 months. Patients receiving adjuvant WBRT had better intracranial control rates; however, this did not lead to improved overall survival. The investigators concluded that in patients with 1 to 3 brain metastases amenable to radiosurgery, SRS alone may be the preferred treatment modality. Retrospective studies were not used to form the recommendation but they also conclude that the addition of WBRT to SRS or surgery is associated with improved local control and distant intracranial control, but not survival. 61 , 62
Lastly, a 2015 meta-analysis by Sahgal et al 60 combined 3 phase III trials to perform a pooled analysis of patients with 1 to 4 brain metastases treated with either SRS alone or SRS + WBRT. The pooled data were individual data obtained from 3 RCTs. 28 , 50, 59 Primary outcomes included survival and local and distant intracranial failure. In total, 364 of the pooled 389 patients met the inclusion criteria and were included in the meta-analysis. Fifty-one percent were treated with SRS alone and 49% were treated with SRS + WBRT. The results showed that patients <50 years of age had a significant survival benefit when SRS was used alone. The median survival for these younger patients was 13.6 months in the SRS only group as opposed to 8.2 months in the SRS and WBRT group (p=0.04). Furthermore, in patients 50 years of age or less, there was no significant difference between the 2 treatment groups with respect to distant brain failure. In older patients, the risk of observed distant failure was higher in the SRS alone cohort. Additionally, patients of any age with a single brain metastases had a lower chance of developing further brain metastases as compared to those patients with 2 to 4 brain metastases (hazard ratio= 0.63). In all patients, SRS and WBRT was associated with a lower hazard of local brain failure than SRS alone (hazard ratio 2.56). Median time to death in the SRS alone versus SRS + WBRT was 10 versus 8.2 months, respectively. The authors concluded that SRS alone is the recommended initial therapy of patients <50 years of age with 1 to 4 brain metastases.
Several Class III studies have addressed the use of SRS alone in patients with > 4 brain metastases and confirmed that overall survival is not different for patients with > 4 brain metastases compared with 1 or 2 to 4 metastases. 63 , 64 In 1 study, patients with total tumor volumes > 7 cc or > 7 metastases had significantly poorer overall survival than patients with smaller volumes or number of metastases. 65 However, when comparing survival according to the RTOG-recursive partitioning analysis (RPA) classifications, patients undergoing SRS appeared to have an improved survival compared with the RTOG historical classification groups. 66 Another retrospective study found that overall survival was predicted more by the volume of brain metastases and distant metastases, rather than the number of metastases. 67 Chang et al64 reached a similar conclusion, in that the overall survival was not significantly different in patients treated with SRS for 1 to 5, 6 to 10, 11 to 15, or >15 brain metastases, with a median survival of 10 months. The overall median progression-free survival was 9 months for thetotal group as opposed to 6 months in patients with >15 lesions (p=0.028). However, patients with more than 15 metastases had a shorter time to progression of new brain metastases.
In summary, compared with surgical resection or radiosurgery alone, WBRT improves intracranial progression-free survival but not overall survival in patients <4 brain metastases. This supports a Level 2 recommendation to not proceed to WBRT in WHO performance status 0-2 patients with <4 brain metastases because, compared with surgical resection or radiosurgery alone, the addition of WBRT improves intracranial progression-free survival but not overall survival. However, local therapy alone is associated with a higher incidence of both local and distant intracranial tumor recurrence, and prospective randomized studies in patients with >4 brain metastases have not been conducted. This supports the following Level 3 recommendation, “Compared with surgical resection or radiosurgery alone, the addition of WBRT is not recommended for patients with >4 brain metastases unless the metastases’ volume exceeds 7 cc, or there are >15 metastases, or the size or location of the metastases are not amenable to surgical resection or radiosurgery.”
Synthesis of Results
WBRT has been a treatment of brain metastases for many years, and RCTs, summarized in Table 2, have evaluated various dose fractionation regimens. These provide Class I evidence that altered dose/fractionation schedules of WBRT do not result in significant differences in median survival, local control or neurocognitive function when compared with “standard” WBRT dose / fractionation such as 30 Gy in 10 daily fractions. The choice of which dose/fractionation scheme to use is based on a combination of patient convenience and life expectancy. There is concern that WBRT delivered with a high dose per fraction, (ie, >4 Gy per fraction) leads to more frequent or severe neurocognitive impairment, although studies of altered fractionation did not incorporate very robust neurocognitive testing.
Relatively few studies, summarized in Table 3, have been done to evaluate the outcomes of WBRT according to the histopathology or molecular status of the primary cancer. One group of patients who may not benefit from immediate WBRT are NSCLC patients with mutant EGFR or ALK-rearranged cancers. Targeted therapy is an option as initial treatment for asymptomatic brain metastases not amenable to SRS, withholding WBRT until the time of intracranial progression. However, mutant EGFR or ALK-rearranged status is also a positive prognostic factor for WBRT response after WBRT. The question remains as to the optimal timing of WBRT, or whether EGFR or ALK status can be used to predict the benefit of WBRT as opposed to other treatment modalities. Outside of lung cancer, few studies have been done that are relevant to this question. Retrospective studies suggest that HER2-positive patients may have improved outcomes following WBRT compared with HER2-negative patients. The role of WBRT, as opposed to SRS, is also controversial in many histologies, but particularly for patients with melanoma. RCTs that are histology- or molecular status-specific are necessary to sort out many of these issues.
An important addition to this guideline is the question regarding the effect of WBRT on neurocognition. Tables 4, 5, and 6 summarize the neurocognitive effects seen with WBRT or PCI. They also summarize the studies whose goal was to ameliorate these effects. Class I data demonstrate that the addition of WBRT to local therapy (SRS or surgery) is associated with an increased risk of significant neurocognitive decline in patients with <4 brain metastases. This decline is apparent as early as 3 months post-RT and can persist in long-term survivors. Class I evidence also exists to support the Level 3 recommendation to utilize memantine for its nonstatistical tendency of neurocognitive protective effects in patients with brain metastases undergoing WBRT. There is lower level evidence suggesting that HA-WBRT may reduce the risk of neurocognitive decline compared with conventional WBRT.
Table 7 summarizes the additional data used to evaluate the effectiveness of WBRT on non-cognitive endpoints, such as progression-free or overall survival. There are RCTs evaluating the use of surgical resection with or without WBRT in the treatment of patients with 1 brain metastasis. Other RCTs evaluated the use of SRS with or without WBRT for patients with 1 to 4 brain metastases. Withholding WBRT during initial treatment is associated with a higher incidence of both local and distant intracranial tumor recurrence but without a detriment to overall survival or performance status. This led to the Level 1 recommendation of surgical resection or SRS alone as the initial treatment for patients with <4 brain metastases. However, there are no Class I studies addressing the benefit of WBRT for patients with more than four brain metastases. Since WBRT improves progression-free survival, this supports a Level 3 recommendation of WBRT following surgical resection or radiosurgery alone.
CONCLUSION AND KEY ISSUES FOR FUTURE INVESTIGATIONS
The use of WBRT has declined over the past 10 years as the use of local and systemic therapies has evolved. A question asked constantly by clinicians is: when is it appropriate to use WBRT? Since the prior publication of this guideline, there have been few studies comparing various dose/fractionation schemes for WBRT. Unless future studies incorporate more sophisticated measures of neurocognitive outcome, there is little need to repeat these studies.
However, technological developments allow WBRT to be delivered with HA to potentially reduce the probability of neurocognitive deficits, which are the most concerning side effect of WBRT. Randomized studies are ongoing to see whether HA does lead to less cognitive impairment without any reduction in intracranial control. Another technological development has been the ability to do an SIB, delivering a higher dose to targeted lesions during a course of WBRT. Prospective trials are ongoing to better support the efficacy of HA and SIB.
The question of when to recommend WBRT, or whether it is of any benefit at all to patients with certain histopathologic or molecular subtypes remains controversial. Recent studies have indicated that the prognosis of brain metastases is more dependent on histopathology or molecular features of the primary cancer than had been appreciated. The role of WBRT as opposed to SRS is also controversial in many histologies, but particularly for patients with melanoma. Whether these histopathology/molecular marker subtypes are both prognostic and predictive of outcomes of WBRT is less clear. Future prospective randomized trials of issues related to WBRT are likely to be more “targeted” to specific populations, such as specific primary cancers or even specific molecular targets. Examples of possible study groups would be HER2-negative breast cancer, EGFR-mutated adenocarcinoma of the lung, or melanoma.
NSCLC cancer patients have been studied in a phase III RCT. 68 Patients with NSCLC and newly diagnosed or progressive brain metastases not amenable to surgical resection or radiosurgery were randomized to either WBRT or supportive care only. There was a broad range of eligibility criteria, but the primary was uncontrolled in approximately two-thirds of patients with extracranial metastases present in >50% of patients and a median Karnofsky Performance Scale score of 60. No significant difference in median survival was found between patients receiving WBRT or supportive care only. The median survival of just 8 to 9 weeks is lower than most prospective studies in brain metastases and raises the question of how patients were selected for the study. In subset analysis, WBRT appeared to provide a survival benefit to patients who were either young, had a controlled primary cancer, or had a low RPA. Nevertheless, this study supports a recommendation of supportive care only for elderly lung cancer patients with a poor Karnofsky Performance Scale score, uncontrolled primary, or progressive systemic disease. Future guidelines will hopefully be able to address this issue in more depth.
There have also been pharmacologic developments to ameliorate the neurocognitive effects of WBRT. The most promising drug is memantine, started early in the course of WBRT and continued for >6 months. Memantine is well tolerated, and few patients will refuse to take it given the risks and benefits. It has been utilized in a North American study of WBRT with HA.53 There is also concern for the potential neurocognitive detriment caused by PCI in patients without known brain metastases. There is an ongoing trial to determine if HA would be beneficial in this patient population ( NRG-CC003 ). This trial randomizes patients with SCLC to PCI to 25 Gy in 10 fractions with or without hippocampal avoidance.
The decision regarding local therapies (SRS and surgery) as opposed to WBRT needs further prospective studies when there are >4 brain metastases. Studies have clearly shown that local therapy is sufficient and reasonable for patients with 1 to 4 brain metastases but the treatment of patients with more numerous metastases still needs to be addressed. Technically, large number of lesions can be treated with SRS, but is that necessarily the appropriate treatment? The main reason to use SRS is partly the convenience to the patient of a short treatment but seems primarily related to concerns of neurocognitive deficit following WBRT and many patients will currently refuse WBRT even when it is recommended. Studies of SRS have not yet documented the neurocognitive effects of SRS, particularly if there are >4 lesions. Further studies to evaluate the timing of WBRT relative to local therapies or systemic therapy would be beneficial to develop patient-specific treatment plans.
Potential Conflicts of Interest
The Brain Metastases Guideline Update Task Force members were required to report all possible conflicts of interest (COIs) prior to beginning work on the guideline, using the COI disclosure form of the AANS/CNS Joint Guidelines Review Committee, including potential COIs that are unrelated to the topic of the guideline. The CNS Guidelines Committee and Guideline Task Force Chair reviewed the disclosures and either approved or disapproved the nomination. The CNS Guidelines Committee and Guideline Task Force Chair are given latitude to approve nominations of task force members with possible conflicts and address this by restricting the writing and reviewing privileges of that person to topics unrelated to the possible COIs. The conflict of interest findings are provided in detail in the companion introduction and methods manuscript.
Disclosures
These evidence-based clinical practice guidelines were funded exclusively by the Congress of Neurological Surgeons and the Tumor Section of the Congress of Neurological Surgeons and the American Association of Neurological Surgeons, which received no funding from outside commercial sources to support the development of this document.
ACKNOWLEDGEMENTS
The authors acknowledge the CNS Guidelines Committee for its contributions throughout the development of the guideline and the AANS/CNS Joint Guidelines Review Committee for its review, comments, and suggestions throughout peer review, as well as Trish Rehring, MPH, CHES, CNS Guidelines Senior Manager, and Mary Bodach, MLIS, Senior Guidelines Specialist, for their assistance. Throughout the review process, the reviewers and authors were blinded from one another. At this time, the guidelines task force would like to acknowledge the following individual peer reviewers for their contributions: Manish Aghi, MD, PhD, Manmeet Ahuwalia, MD, Sepideh Amin-Hanjani, MD, Edward Avila, MD, Maya Babu, MD, MBA, Kimon Bekelis, MD, Priscilla Brastianos, MD, Paul Brown, MD, Andrew Carlson, MD, MS, Justin Jordan, MD, Terrence Julien, MD, Cathy Mazzola, MD, Adair Prall, MD, Shayna Rich, MD, PhD, Arjun Sahgal, MD, Erik Sulman, MD, May Tsao, MD, Michael Voglebaum, MD, Stephanie Weiss, MD, and Mateo Ziu, MD.
Figure 1 PRISMA Flow Diagram
TABLES
Table 1. Search Strategies
|
PUBMED (NLM), searched on February 3-4, 2016:
|
|
Step 1: Brain Neoplasms [Mesh]
|
|
Step 2: (brain OR brainstem OR intracranial) AND (cancer OR tumor* OR tumour* OR neoplasm*) [TIAB]
|
|
Step 3: #1 OR #2
|
|
Step 4: Neoplasm Metastasis [Mesh]
|
|
Step 5: (brain OR brainstem OR intracranial) AND (Metastas*) [TIAB]
|
|
Step 6: #4 OR #5
|
|
Step 7: #3 AND #6
|
|
Step 8: Brain neoplasms/secondary [Mesh]
|
|
Step 9: #7 OR #8
|
|
Step 10: Cranial irradiation [Mesh]
|
|
Step 11: WBRT [TIAB]
|
|
Step 12: “whole brain” [TIAB] AND (radiotherap* OR radiation OR radiation therap* OR irradiation) [TIAB]
|
|
Step 13: #10 OR #11 OR #12
|
|
Step 14: #9 AND #13
|
|
Step 15: #14 AND English [Lang]
|
|
Step 16: (animals [MeSH] NOT humans [MeSH]) OR case reports [PT] OR review [PT] OR comment [PT] OR letter [PT] OR editorial [PT] OR addresses [PT] OR news [PT] OR “newspaper article” [PT]
|
|
Step 17: #15 NOT #16
|
|
Step 18: #17 AND ("1990/10/01"[PDAT] : "2015/12/31"[PDAT])
|
|
Embase, searched on February 3-4, 2016:
|
|
Step 1: ‘Brain tumor’/exp
|
|
Step 2: ((brain OR brainstem OR intracranial) NEAR/3 (cancer OR tumor* OR tumour* OR neoplasm*)):ab, ti
|
|
Step 3: #1 OR #2
|
|
Step 4: ‘brain metastasis’/exp
|
|
Step 5: ((brain OR brainstem OR intracranial) NEXT/3 metastas*):ab,ti
|
|
Step 6: #4 OR #5
|
|
Step 7: #3 AND #6
|
|
Step 8: ‘brain radiation’/exp
|
|
Step 9: WBRT:ab,ti
|
|
Step 10: (‘whole brain’ NEXT/3 (radiation OR radiotherapy* OR irradiation)):ab,ti
|
|
Step 11: #8 OR #9 OR #10
|
|
Step 12: #7 AND #11
|
|
Step 13: Limits: English, humans, 1990-2015, article OR conference paper NOT case report
|
|
COCHRANE, searched on February 3-4, 2016 :
|
|
Step 1: MeSH descriptor: [Brain Neoplasms] explode all trees
|
|
Step 2: ((brain OR brainstem OR intracranial) NEAR/3 (cancer OR tumor* OR tumour* OR neoplasm*)):ti,ab,kw
|
|
Step 3: #1 OR #2
|
|
Step 4: MeSH descriptor: [Neoplasm Metastasis] explode all trees
|
|
Step 5: ((brain OR brainstem OR intracranial) NEAR/3 Metastas*):ti,ab,kw
|
|
Step 6: #4 OR #5
|
|
Step 7: #3 AND #6
|
|
Step 8: MeSH descriptor: [Brain neoplasms/secondary]
|
|
Step 9: #7 OR #8
|
|
Step 10: MeSH descriptor: [Cranial irradiation] explode all trees
|
|
Step 11: WBRT:ti,ab,kw
|
|
Step 12: (‘whole brain’ NEXT/3 (radiation OR radiotherapy* OR irradiation)):ti,ab,kw
|
|
Step 13: #10 OR #11 OR #12
|
|
Step 14: #9 AND #13
|
|
Step 15: Filtered 1990-2015
|
Table 2. Outcomes of different dose/fractionation schedules of whole brain radiation therapy
|
Author (Year)
|
Description of Study
|
Data Class
|
Conclusions
|
|
Sayed21 (2015)
|
Study description
Prospective nonrandomized study at 1 center to compare 2 WBRT regimens for differences in response and overall survival.
Patient population
93 patients with MRI scan with >3 brain metastases, good performance status.
Treatment regimen
G1: 20 Gy in 4 Gy fractions (n = 54)
G2: 30 Gy in 3 Gy fractions (n = 39)
|
III
|
Results
Median survival
G1: 9 months
G2: 10 months
(p = 0.02)
MRI response at 3 months (partial response or stable)
G1: 85%
G2: 87%
(p = NS)
Author’s conclusions
No significant difference in response or overall survival. Shorter fractionation beneficial to patients with RPA 2 (less time spent in treatment and little concern for late toxicity) and to radiation facilities (quicker throughput).
Comments and conclusions
No neurocognitive testing. Designated as Class III because it was a very small prospective study with “assignment” to 1 of 2 dose schedules. Statistical rationale for the accrual goal not given.
|
|
Saha et al20 (2014)
|
Study description
RCT in multiple centers comparing outcome of 2 WBRT regimens.
Patient population
56 patients with radiologic diagnosis of brain metastases on MRI, good performance status
Treatment regimen
G1: 20 Gy in 4 Gy fractions (n = 26)
G2: 30 Gy in 3 Gy fractions (n = 30)
|
III
|
Results
Median survival
G1: 26 weeks
G2: 29 weeks
(p = 0.955)
MRI response at 3 months (complete or partial response or stable)
G1: 81%
G2: 93%
Author’s conclusions
No significant difference in response or overall survival. 20 Gy in 5 fractions recommended for patients with poor performance status, 30 Gy in 10 fractions for patients with good performance status.
Comments and conclusions
No neurocognitive testing. No significant difference in improvement in ADL between 2 arms, but ADL of both groups improved post-WBRT. Designated as Class III since the patient numbers are small and could account for the nonsignificant finding. Statistical rationale for the accrual goal not given.
|
|
Graham et al19 (2010)
|
Study description
RCT in cooperative group (ECOG) to compare intracranial control rate and QOL of 2 WBRT schemes.
Patient population
113 patients with good performance status; stable, absent, or concurrent presentation of extracranial disease
Treatment regimen
G1: 40 Gy in 2 Gy fx BID (n = 57)
G2: 20 Gy in 5 Gy fx (n = 56)
|
I
|
Results
Median survival
G1: 6.1 months
G2: 6.6 months
(p = NS)
Intracranial progression
G1: 44%
G2: 64%
(p = 0.03)
Author’s conclusions
Intracranial disease control was improved and QOL maintained with 40 Gy in 20 twice-daily fractions. Authors recommend this dose/fractionation for patients with better prognosis.
Comments and conclusions
The dose/fractionation regimen was not a significant factor affecting overall survival on MVA. Significant factors for improved survival on MVA were resection, supratentorial location, absent extracranial metastases, younger age. QOL and cognitive function outcomes similar in both groups. Mean scores of QOL and cognitive function were stable to improved in most patients during the 6-9 months following treatment.
|
|
Davey et al5 (2008)
|
Study description
RCT at 2 centers to compare overall survival following accelerated and conventional hypofractionated WBRT.
Patient population
90 patients with radiologic features of brain metastases on CT or MRI. Good performance status, life expectancy >6 weeks.
Treatment regimen
G1: 20 Gy/5 daily fractions (n = 45)
G2: 40 Gy/20 fractions/twice daily (n = 45)
|
I
|
Results
Median survival
G1: 19.1 weeks
G2: 19.1 weeks
(survival curves: log-rank; p = NS)
Median time to treatment for intracranial relapse
G1:14 weeks
G2: 32 weeks
(p = 0.03)
Author’s conclusions
Although accelerated WBRT may improve intracranial control, this did not lead to improved overall survival.
Comments and conclusions
No QOL or neurocognitive testing. Favorable prognostic factors on MVA were low RPA class and colorectal pathology.
|
|
Murray et al9 (1997)
|
Study description
RCT by cooperative group (RTOG) comparing accelerated hyperfractionated WBRT with standard fractionation.
Patient population
429 patients with brain metastases measurable by CT or MRI scans, Karnofsky scale score ≥70, neurologic function class of 1-2.
Treatment regimen
G1: 30 Gy/10 fractions/daily (n = 213)
G2: 54.4 Gy/34 fractions/twice daily (n = 216)
|
I
|
Results
Median survival
G1: 4.5 months
G2: 4.5 months
(p = NS)
# pts with recurrence/progression
G1: 109/124 (88%)
G2: 105/118 (89%)
(p value not reported)
Median time to recurrence / progression
G1: 11 weeks
G2: 10 weeks
(p value not reported)
Author’s conclusions
54.4 Gy in 34 fractions not recommended.
Comments and conclusions
No neurocognitive testing. 54.4 Gy delivered as 32 Gy in 20 fractions/twice daily followed by 24.4 Gy boost (visible lesion with 2-cm margin) in 14 fractions/twice daily. Age, performance status, extent of metastatic disease, and status of primary were prognostic factors.
|
|
Priestman et al10 (1996)
|
Study description
RCT at 25 institutions comparing 2 WBRT regimens
Patient population
544 patients with symptomatic brain metastases by CT scan or unequivocal radioisotope scan, or an intracranial biopsy. Required stable dose dexamethasone over week prior to randomization, WHO performance status of 0-3, neurologic status <4 by modified MRC scale
Treatment regimen
G1: 12 Gy/2 fractions (n = 274)
G2: 30 Gy/10 fractions (n = 270)
|
I
|
Results
Median survival
G1: 77 days
G2: 84 days
(p = 0.04 for entire survival curve, no difference in median survival)
Author’s conclusions
For majority of patients, no advantage to longer courses of radiation therapy.
Comments and conclusions
No neurocognitive testing. Small improvement in survival with longer course but not thought by authors to be clinically meaningful. Might recommend longer course in small number of patients with good prognosis (female gender, age <60 years, breast primary, solitary brain metastasis, dexamethasone ≤8 mg/day, WHO performance status <3).
|
|
Chatani et al3 (1994)
|
Study description
RCT evaluating 2 different WBRT regimens in patients with normal (<250 U/L) vs high LDH
Patient population
162 patients with lung cancer (stratified for small vs nonsmall) with CT brain scan.
Treatment regimen
Normal LDH:
G1: 30 Gy/10 fractions (n = 46)
G2: 50 Gy/20 fractions with field reduction after 30 Gy if possible (n = 46)
High LDH:
G3: 30 Gy/10 fractions (n = 35)
G4: 20 Gy/5 fractions (n = 35)
|
II
|
Results
Median survival
G1: 5.4 months
G2: 4.8 months
(p = NS)
G3: 3.4 months
G4: 2.4 months
(p = NS)
Author’s conclusions
LDH is important prognostic factor. 30 Gy/10 fractions recommended.
Comments and conclusions
No neurocognitive testing. RCT but designated as class II and the patient numbers were small, with no clear inclusion criteria beyond “lung cancer.”
|
|
Sause et al11 (1993)
|
Study description
Cooperative group (RTOG) phase I/II trial of accelerated fractionation
Patient population
Patients eligible had controlled or absent primary with metastases than brain stable, or only brain metastases with primary uncontrolled.
Treatment regimen
G1: 32 Gy in 1.6 Gy fractions + boost to 48.0 Gy] (n = 62)
G2: 32 Gy in 1.6 Gy fractions + boost to 54.4 Gy] (n = 115)
G3: 32 Gy in 1.6 Gy fractions + boost to 64.0 Gy] (n = 104)
G4: 32 Gy in 1.6 Gy fractions + boost to 70.4 Gy] (n = 53)
Fractions administered twice daily
|
II
|
Results
Median survival
G1: 4.2 months
G2: 5.2 months
G3: 4.8 months
G4: 6.4 months
(p = NS)
Author’s conclusions
Nonsignificant improvement in survival in higher dose arms was taken as an encouraging result.
Comments and conclusions
No neurocognitive testing. Used as basis for subsequent RTOG study.9 Designated as class II since it was a phase I/II randomized phase II study within cooperative group (RTOG).
|
|
Haie-Meder et al6 (1993)
|
Study description
RCT at 3 institutions comparing 2WBRT treatment regimens
Patient population
216 patients with lung, breast, head and neck, or unknown primaries. Diagnosed by CT scan. Age <71 years. Ineligible if Karnofisky scale score <20 or life expectancy <1 month
Treatment regimen
G1: 18 Gy/3 fractions (n = 110)
G2: 18 Gy/3 fractions; 4 weeks later a second identical course or 25 Gy/10 fractions (n = 106)
|
I
|
Results
Median survival
G1: 4.2 months
G2: 5.3 months
(p = NS)
Author’s conclusions
No difference in overall survival or neurologic response or incidence in complications. A radiation schedule as short as 18 Gy in 3 fractions as good as longer radiation schedules. No neurologic complications occurred among 45 patients living >12 months
Comments and conclusions
Investigators could decide on whether G2 received 18 or 25 Gy in the second course- shortest regimen recommended if poor general or neurologic status. Methods of assessing neurocognitive function in follow-up were not clearly described. Two clinical factors predictive of poor survival were presence of multiple brain metastases and/or extracranial metastases.
|
|
Komarnicky et al7 (1991)
|
Study description
RCT by cooperative group (RTOG) evaluating role of misonidazole combined with WBRT
Patient population
859 patients with measurable disease on CT, 18-75 years of age, Karnofsky scale score ≥40, able to work
Treatment regimen
G1: 30 Gy/10 fractions (n = 193)
G2: 30 Gy/6 fractions (n = 200)
G3: 30 Gy/6 fractions + MISO (n = 196)
G4: 30 Gy/10 fractions + MISO (n = 190)
|
I
|
Results
Median survival
G1: 4.5 months
G2: 4.1 months
G3: 3.1 months
G4: 3.9 months
(p = NS)
# of pts retreated for BM after protocol therapy
G1: 54/179 (30%)
G2: 54/180 (30%)
G3: 33/173 (19%)
G4: 54/163 (33%)
(p = NS)
Author’s conclusions
Recommended treatment was 30 Gy in 10 fractions, without misonidazole
Comments and conclusions
No neurocognitive testing. Approximately one-third of patients died of uncontrolled metastases, suggesting the need for more effective therapy.
|
|
Chatani et al4 (1985)
|
Study description
RCT at a single institution
Patient population
69 consecutive patients with metastases from lung cancer
Treatment regimen
G1: 30 Gy/10 fractions (n = 35)
G2: 50 Gy in 20 fractions (n = 34)
|
II
|
Results
Median survival
G1: 4 months
G2: 3 months
(p = NS)
Survival at 6 months
G1: 42%
G2: 14%
(p < 0.05)
Author’s conclusions
Performance status and LDH were the factors influencing 6-month survival
Comments and conclusions
No neurocognitive testing. Designated as Class II due to small numbers and was limited to lung cancer.
|
|
Kurtz et al8 (1981)
|
Study description
RCT by cooperative group (RTOG)
Patient population
309 patients (255 evaluable) from 31 participating institutions. Ineligible if evidence of other sites of metastatic disease or progressive untreated primary, or poor neurologic function
Treatment regimen
G1: 30 Gy/10 fractions (n = 130)
G2: 50 Gy/20 fractions (n = 125)
|
I
|
Results
Median survival
G1: 18.2 weeks
G2: 16.9 weeks
(p = NS)
# pts with recurrence/progression in patients with information available
G1: 109/124 (88%)
G2: 105/118 (89%)
(p value not reported)
Author’s conclusions
30 Gy in 10 fractions as effective as 50 Gy.
Comments and conclusions
Excluded patients with evidence of extracranial metastases, uncontrolled primaries, or poor neurologic function. 21% of patients in 50 Gy arm unable to complete therapy. No neurocognitive testing. Authors recommended 20-30 Gy in 5-10 fractions.
|
|
Borgelt et al2 (1981)
|
Study description
Two large (>900 patients in each study) national RCTs by cooperative group study (RTOG) with optional randomization to very short regimens at small number of institutions. This study is analysis of patients randomized at 4-6 centers that had very short regimens open.
Patient population
Ineligible if lesions too numerous or symptoms too vague to allow for adequate follow-up or assessment.
First RCT: 155 patients randomized at 6 institutions
Second RCT: 78 patients randomized at 4 institutions
Treatment regimen
First RCT:
30 Gy/10 fractions/2 wks (n = 233)
30 Gy/15 fractions/3 wks (n = 217)
40 Gy/15 fractions/3 wks (n = 233)
40 Gy/20 fractions/4 wks (n = 227)
10 Gy/single fraction: option in 6 institutions (n = 26)
Second RCT:
20 Gy/5 fractions/1 wk (n = 31)
12 Gy in 2 fractions (n = 33)
Analysis by group
First RCT:
G1: 10 Gy/1 fraction
G2: 30-40 Gy over 2-4 weeks
Second RCT:
G3: 12 Gy/2 fractions
G4: 20 Gy over 1 week
|
I
|
Results
Median survival
First RCT:
G1: 15 weeks
G2: 21 weeks
(survival curves: log-rank; p = NS)
Second RCT:
G3: 13 weeks
G4: 12 weeks
(survival curves: log-rank; p = NS)
Median time to progression (measured by deterioration in neurologic function ):
First RCT:
Initial NF 1: G1: 9 wks; G2: 14 wks
Initial NF 2: G1: 9 wks; G2: 10 wks
Initial NF 3: G1: 7 wks; G2: 12 wks (Cox’s model; p = 0.07)
Second RCT:
Initial NF 1: G3: 9 wks; G4: 10 wks
Initial NF 2: G3: 11 wks; G4: 8 wks
Initial NF 3: G3: 3 wks; G4: 3 wks (Cox’s model; p = NS)
Authors’ conclusions
Response of patients receiving the ultra-rapid treatment (10-12 Gy in 1-2 fractions) as assessed by the percent who had improvement in neurologic function, was comparable to that of patients receiving the more protracted schedules. Promptness of neurologic function improvement, treatment morbidity, and median survival were also comparable to those of patients receiving the more protracted courses. However, the duration of improvement, time to progression of neurologic status and rate of complete disappearance of neurologic symptoms were generally less for patients treated with ultrarapid treatment. Ultrarapid treatment may not be as effective as higher dose schedules in the palliation of brain metastases.
Comments and conclusions
No neurocognitive testing. Large cooperative group RCT but relatively small numbers of patients in the second RCT testing ultrarapid treatment.
|
|
Borgelt et al33 (1980)
|
Study description
2 RCT by cooperative group (RTOG) to study effectiveness of different WBRT dose fractionation schemes on palliation.
Patient population
First RCT 993 (910 evaluable) and second RCT 1001(902 evaluable) patients with brain metastases established by clinical symptoms, EEG, radioisotope brain scan, arteriogram, pneumoencephalogram, or biopsy. Patients excluded if lesions too numerous or symptoms too vague to allow for adequate follow-up or assessment.
Treatment regimen
First RCT:
G1: 30 Gy/10 fractions/2 wks (n = 233)
G2: 30 Gy/15 fractions/3 wks (n = 217)
G3: 40 Gy/15 fractions/3 wks (n = 233)
G4: 40 Gy/20 fractions/4 wks (n = 227)
Second RCT:
G1: 20 Gy/5 fractions/ 1 wk (n = 447)
G2: 30 Gy/10 fractions/ 2 wks (n = 228)
G3: 40 Gy/15 fractions/ 3 wks (n = 227)
|
I
|
Results
Median survival
First RCT: 18 weeks. No significant difference between G1-4 (range 16-20 wks)
Second RCT: 15 weeks. No significant difference between G1-3 (range 14-15 wks)
Brain metastases as cause of death
First RCT: 49%. No significant difference between G1-4 (range 46-54%)
Second RCT: 31% No significant difference between G1-3 (range 25-33%)
Palliation of neurologic symptoms
Relief in 60-90% of patients with no significant difference between studies
Improvement in neurologic function at 2 weeks
First RCT:
G1: 55%
G2-4: 43%
(p = 0.06)
Second RCT:
G1: 64%
G2-3: 54%
(p = 0.01)
Author’s conclusions
All treatment schedules were comparable with respect to frequency of improvement, duration of improvement, time to progression, survival, and palliation. Important prognosticators of response included initial neurologic function and general performance status. Administration of steroids during irradiation favored more rapid improvement
Comments and conclusions
The administration of steroids was not controlled in either study. Results by treatment regimens not presented separately. Primary site (lung vs breast vs other) had no influence on palliative benefit of WBRT. Palliation reported sooner in shorter WBRT regimens but reporting bias suspected. Relatively small numbers of patients in the second RCT testing ultrarapid treatment. No neurocognitive testing.
|
ADL, activities of daily living; BID, twice daily; CT, computed tomography; ECOG, Eastern Cooperative Oncology Group; Gy, Gray; LDH, lactate dehydrogenase; MRC, Medical Research Council; MRI, magnetic resonance imaging; MVA, multivariate analysis; QOL, quality of life; RCT, randomized controlled trial; RPA, recursive partitioning analysis; WBRT, whole brain radiation therapy; WHO, World Health Organization.
Table 3. Effect of histology of primary cancer on outcomes of whole brain radiation therapy
|
Author (Year)
|
Description of Study
|
Data Class
|
Conclusions
|
|
Lee et al30 (2012)
|
Study description
Single institution, retrospective review of impact of EGFR mutation in patients with NSCLC brain metastases treated with WBRT in terms of RPFS and OS
Patient population
43 patients with NSCLC (40 adenocarcinoma, 1 adenosquamous carcinoma, 2 poorly differentiated carcinoma)
EGFR-positive: 30 patients with EGFR mutation (15 with exon 19 deletions, 15 with exon 21 L858R point mutation);
EGFR-negative: 13 patients with EGFR wild-type
Treatment regimen
43 patients underwent WBRT (30-40 Gy in 10-20 fractions, 40% of patients had additional local boost up to 50-60 Gy).
EGFR tyrosine kinase inhibitor (TKI) given to 50% of EGFR-positive and 69% of EGFR-negative patients.
|
III
|
Results
Median follow-up 15 months
Radiographic response to RT
Overall 70% radiographic response rate to RT
EGFR-positive: 80%
EGFR-negative: 46 (p = 0.037)
Multivariate analysis of radiographic response
EGFR mutation was only predictor for treatment response (odds ratio: 4.67, 95% CI; p = 0.032)
Median intracranial RPFS
Overall 18 months (95% CI: 8.33-27.68)
EGFR-positive: 21 months
EGFR-negative: 12 months (p = 0.009)
Multivariate analysis for RPFS
EGFR mutation (p = 0.025) and RPA class ( p = 0.026) were 2 predictors for longer RPFS
Overall survival
Median OS 15 months (95% CI: 9.61-20.39 months)
Univariate analysis showed that EGFR mutations ( p = 0.061) and performance status (p = 0.076) had a trend to predict OS.
Author’s conclusion
Mutant EGFR in NSCLC brain metastasis patients is an independent prognostic factor for better treatment response and longer intracranial RPFS following WBRT
Comments and conclusions
This is a retrospective case series (class III) of patients with brain metastasis from NSCLC treated with WBRT, which found mutant EGFR as a positive prognostic factor for treatment response after WBRT. EGFR TKI given to more than half of these patients and difficult to know how this impacted results. EGFR TKI should not be given to patients known to be EGFR wild-type, since it has been shown in other settings to be associated with poor outcome.
|
|
Gow et al31 (2008)
|
Study description
Single institution, retrospective case series of patients with brain metastases from lung adenocarcinoma treated with WBRT, evaluating the role of EGFR mutation status in response to WBRT and survival
Patient population
63 patients patient with brain metastases from lung adenocarcinoma treated with WBRT
EGFR-positive: Positive EGFR mutations (n = 46)
EGFR-negative: Wild-type EGFR (n = 17)
Treatment regimen
63 patients with NSCLC brain metastases received WBRT (30-35 Gy in 15 to 18 fractions); 18 patients received gefitinib treatment (either before or during WBRT treatment).
Pertinent methods of study technique
Univariate and logistic regression models were used to test predictive factors associated with clinical response; log-rank test and cox regression were used to identify factors affecting survival
|
III
|
Results
Clinical response to WBRT
Overall response rate 46%
EGFR-positive: 54%
EGFR-negative: 24% (p = 0.045)
Both EGFR expression and EGFR tyrosine kinase inhibitor administration were independently associated with response to WBRT (p = 0.034 and p = 0.029, respectively)
Survival with WBRT
Median survival was 14.7 months (95% CI, 7.5-21.9 months)
Better OS in responders vs nonresponders to WBRT (20.7 vs 6.6 months, p = 0.017).
On univariate analysis, RPA class (p = 0.025), KPS (p = 0.013), and absence of extracranial metastases (p = 0.005) were significant prognosticators for overall survival.
EGFR mutation (p = 0.131) and administration of EGFR TKI during WBRT (p = 0.121) showed a trend but no significant correlation with survival.
Author’s conclusion
EGFR mutation and EGFR TKI administration during WBRT are independent predictors of response to WBRT in brain metastases from lung adenocarcinoma.
Comments and conclusion
This retrospective case series (class III) found mutant EGFR expression and TKI administration were predictive of improved response to WBRT, with a trend to improved overall survival but not statistically significant. All patients received WBRT but a small number also received systemic therapy with gefitinib, representing a heterogeneous treatment population.
|
|
Wolstenholm et al32 (2008)
|
Study description
Single institution, retrospective case series examining the influence of HER2 status on outcome of patients with brain metastases from breast cancer who received WBRT
Patient population
181 patients with breast cancer metastasis and known HER2 status receiving WBRT
HER2+ (n=88)
HER2- (n=93)
Treatment regimen
WBRT regimens included 20 Gy in 5 fractions or 30 Gy in 10 fractions (5 and 2 patients in the HERR2+ and HER2- groups respectively received surgery as primary treatment followed by WBRT, and 11 and 1 patients in the HER2+ and HER2- groups respectively received stereotactic radio surgery (18-22 Gy at the 90-100%) in addition to WBRT.
Heterogeneous chemotherapy regimens; trastuzumab treatment in 53 HER2+ patients.
Pertinent methods of study technique
Univariate and multivariate Cox regression analysis of prognostic factors; Kaplan-Meier survival analysis with log-rank test
|
III
|
Results
Median survival after WBRT
HER2-: 8 months
HER2+: 4 months
p=0.008
Prognostic factors
8 patients (4% of entire study population) had solitary brain metastases, with significantly improved survival compared to multiple brain metastases (p=0.005); 6 of these patients were HER2+
On univariate analysis performance status was significant predictor of longer survival (p=0.01)
On multivariate analysis HER2 status was an independent prognostic factor (p=.02)
Author’s conclusion
Improved median survival in patients with HER2+ status following WBRT, which could be attributed to a more aggressive approach to their management with combined cytotoxic chemotherapy and ongoing trastuzumab.
Comments and conclusions
This is a retrospective study (Class III) with no comparison group, with a heterogeneous mix of treatments in addition to WBRT and varied chemotherapy regimens, including use of trastuzumab in a portion of the HER2+ patients.
|
|
Sundstrom et al29 (1998)
|
Study description
Single institution, retrospective review of patients treated with WBRT for brain metastases diagnosed by CT or MRI with minimum midline dose to the whole brain of at least 25 Gy.
Patient population
Breast cancer (n=19)
Lung cancer (n=35)
Renal cell (n=9)
Melanoma (n=6)
Other (n=6)
Extra-cranial metastases
Breast: 17/19
Lung: 6/35
Renal cell: 5/9
Melanoma: 4/6
Other: 5/6
Treatment regimens
WBRT mean dose 30 Gy (range 25–40 Gy) in 1.8– 3 Gy fractions
|
III
|
Results
Median survival by primary tumor type
Breast cancer: 7 months (range 1–62 months)
Lung cancer: 4 months (range 1–21 months)
Renal cell: 4 months (range 2–34 months)
Melanoma: 3 months (range 1–6 months)
Other: 4 months (range 1–9 months)
Survival curves: P-value not reported
Median time to recurrence of brain metastases Not reported
Tumor control, functional performance, cause of death, adverse events Not reported by histology
Author’s conclusions
Approximately two-thirds of the patients experienced a relief in symptoms allowing a reduction in the dose of corticosteroid medication, which clearly supports the use of whole-brain radiotherapy as a palliative treatment.
Comments and conclusions
Designated Class III since numbers too small to allow meaningful statistical comparison between histologies.
|
|
Borgelt et al33 (1980) and Borgelt et al 2 (1981)
|
Study description
2 RCT by cooperative group (RTOG) to study effectiveness of different WBRT dose fractionation schemes on palliation.
Patient population
First RCT 993 (910 evaluable) and second RCT 1001 (902 evaluable) patients with brain metastases established by clinical symptoms, EEG, radioisotope brain scan, arteriogram, pneumoencephalogram, or biopsy.
Stratified by site of primary lesion: lung vs breast vs other, and presence or absence of metastases to sites other than brain; pPatients excluded if lesions too numerous or symptoms too vague to allow for adequate follow-up or assessment.
Treatment regimen
First RCT:
G1: 30 Gy/10 fractions/2 wks (n = 233)
G2: 30 Gy/15 fractions/3 wks (n = 217)
G3: 40 Gy/15 fractions/3 wks (n = 233)
G4: 40 Gy/20 fractions/4 wks (n = 227)
Second RCT:
G1: 20 Gy/5 fractions/ 1 wk (n = 447)
G2: 30 Gy/10 fractions/ 2 wks (n = 228)
G3: 40 Gy/15 fractions/ 3 wks (n = 227)
|
II
|
Results
Median survival
First RCT: 18 weeks. No significant difference between G1-4 (range 16-20 wks)
Second RCT: 15 weeks. No significant difference between G1-3 (range 14-15 wks)
Brain metastases as cause of death
First RCT: 49%. No significant difference between G1-4 (range 46-54%)
Second RCT: 31% No significant difference between G1-3 (range 25-33%)
Primary site
60% of patients had lung primaries.
Lung cancer patients more likely to have brain as only site of metastases; primary site had no influence on response to WBRT.
Time to progression longer for breast cancer patients.
Median survival for breast cancer patients longer than for lung cancer patients (21 weeks vs 16 wks, p < 0.001). This survival difference between breast and lung cancer not seen in nonambulatory patients.
Author’s conclusions
All treatment schedules were comparable with respect to frequency of improvement, duration of improvement, time to progression, survival, and palliation. Important prognosticators of response included initial neurologic function and general performance status. Administration of steroids during irradiation favored more rapid improvement
Comments and conclusions
Primary site (lung vs breast vs other) had no influence on palliative benefit of WBRT. The administration of steroids was not controlled in either study. Palliation reported sooner in shorter WBRT regimens but reporting bias suspected. Relatively small numbers of patients in the second RCT testing ultrarapid treatment. Designated class II since results by treatment regimens not presented separately by histology.
|
EGFR, epidermal growth factor receptor; Gy, Gray; KPS, Karnofsky Performance Scale; NSCLC, non–small cell lung cancer; OS, overall survival; RCT, randomized controlled trial; RPFS, radiologic progression-free survival; TKI, tyrosine kinase inhibitor; WBRT, whole brain radiation therapy.
Table 4. Neurocognitive outcomes of prophylactic cranial irradiation versus no prophylactic cranial irradiation for patients without brain metastases
|
Author and Year
|
Description of Study
|
Data Class
|
Conclusions
|
|
Wolfson et al43 (2011)
|
Study description
Secondary endpoint of multi-institutional phase II RCT
SCLC histology (N = 264)
Testing different PCI RT schedules for patients with SCLC in complete remission after induction therapy.
Treatment regimens
G1: 25 Gy in 10 fractions (n = 131)
G2: 36 Gy in 18 fractions (n = 67)
G3: 36 Gy in 24 fractions, 1.5 Gy BID (n = 66)
Randomization to 25 Gy vs 36 Gy, then secondary randomization to G2 vs G3.
ND defined as a significant decrease at 12 months in at least one neurocognitive test (HVLT, COWAT, or TMT-A and -B) from baseline regardless of brain metastases.
|
II
|
Results
Statistically significant differences for COWAT (p = 0.03) and TMT-A (adjusted p = 0.03) testing at baseline among the 3 groups.
Proportion with ND (regardless of brain metastases) at 12-months:
G1: 62%
G2: 85%
G3: 89%
Significant difference in ND between G1 and G2/3 (p = 0.03)
Proportion with ND without brain metastases at 12-months:
G1: 60%
G2: 85%
G3: 89%
Significant difference in ND between G1 and G2/3 (p = 0.02)
Logistic regression model for ND without brain metastases at 12 months showed significantly higher risk with 36 Gy (p = 0.03) and older age (p = 0.005)
Author’s conclusion
Due to increased risk of ND with 36 Gy PCI, 25 Gy PCI remains standard of care for this patient population
Comments and conclusions
Formal neurologic testing within prospective trial indicating that ND increased with increasing WBRT dose, and there was no beneficial neurocognitive effect to BID fractionation. Designated as Class II since neurologic decline was a secondary endpoint.
|
|
Le Péchoux et al44 (2011)
|
Study description
Secondary endpoint of international multi-institutional phase III RCT for SCLC histology.
Testing different PCI RT schedules for patients with limited SCLC in complete remission after induction therapy
Treatment regimens
G1: 25 Gy in 10 fractions (n = 360)
G2: 36 Gy in 18 daily fractions or 24 fractions of 1.5 Gy BID (n = 360)
|
II
|
Results
Proportion of patients with abnormal QoL-cognitive functioning (scale <75) at baseline (N = 667 with baseline data available)
G1: 23%
G2: 25%
Proportion of patients with abnormal QoL-cognitive functioning (scale <75) at 24-months (n = 140)
G1: 41%
G2: 46%
Proportion of patients with abnormal LENT-SOMA intellectual functioning at 24-months (n = 144)
G1: 20%
G2: 28%
G1 and G2 showed a similar, mild deterioration across time in communication deficit, weakness of legs, intellectual deficit and memory. This deterioration over time was statistically significant (p < 0.005).
Author’s conclusion:
Patients should be informed of the potential neurologic and neurocognitive deficits, as well as the benefit of PCI on survival and the incidence of brain metastases. 25 Gy remains the standard of care for PCI for limited SCLC.
Comments and conclusions
Large RCT in cooperative group using validated QOL tools. Designated as class II since neurologic decline was a secondary endpoint.
|
|
Sun et al45 (2011)
|
Study description
Secondary endpoint of US multi-institutional phase III RCT in NSCLC histology
PCI vs no PCI for patients with stage IIIA/B NSCLC without disease progression after definitive therapy.
Treatment regimens
No PCI (n= 163)
PCI 30 Gy in 15 fractions (n = 177)
Accrual was 340 eligible patients out of planned 1058 (trial closed early due to poor accrual).
|
III
|
Results
Baseline neurocognitive results not reported.
Baseline used for per patient measurement of decline
Proportion with significant deterioration in HVLT-IR at 1 year (n = 90)
Control: 7% PCI: 26% (adjusted p = 0.03)
Proportion with significant deterioration in HVLT-DR at 1 year (n = 90)
Control: 5% PCI: 32% (adjusted p = 0.008)
Proportion with deterioration in MMSE score as defined by reliable change index (n = 95)
Control: 18% PCI: 23% (p = NS)
Authors conclusion
No significant differences in global cognitive function (MMSE) or QOL after PCI, but there was a significant decline in memory (HVLT) at 1 year.
Comments and conclusions
This was designated as class III given that it closed with only approximately one third of planned accrual, perhaps accounting for the lack of significant differences.
|
|
Slotman et al46 (2009)
|
Study description
Secondary endpoint of European multi-institutional phase III RCT
SCLC histology, extensive stage with response to induction therapy
Treatment regimen
PCI (n = 143)
No PCI (n = 143)
Most common PCI dose fractionation regimens:
20 Gy in 5 fractions (62%)
30 Gy in 10 fractions (16%)
30 Gy in 12 fractions (6%)
25 Gy in 10 fractions (5%)
HRQOL measured with EORTC Quality of Life Questionnaire C30 (EORTC-QLQ-C30) and EORTC QLQ Brain Cancer Module (EORTC-QLQ-BN20)
268 of 286 with baseline scores available.
|
II
|
Results
Proportion with worsened global health status (≥ 20-point decline) at 3 months (n = 188)
PCI: 34.7%
No PCI: 22.2% (p = NS)
Proportion with worsened cognitive functioning (≥ 20-point decline) at 3 months (n = 188)
PCI: 22.4%
No PCI: 10% (p = NS)
Mean difference in cognitive functioning score at 3 months between arms (No PCI – PCI) of 8.8 points (below significance definition of ≥10 points)
Authors conclusions:
PCI should be offered to all responding ED SCLC patients. Patients should be informed of the potential adverse effects from PCI.
Comments and conclusions
The largest mean difference between the 2 arms was observed for fatigue and hair loss. The impact of PCI on global health status as well as on neurocognitive functioning scores was more limited. Designated as Class II since change in cognitive function was a secondary endpoint.
|
|
Gregor et al47 (1997)
|
Study description
Secondary endpoint of UKCCCR and EORTC multi-institutional phase III RCT of SCLC histology.
Included patients without brain metastases with complete remission after induction therapy
Treatment regimen
PCI (n = 120)
No PCI (n = 194)
Most common PCI regimens were 30 Gy in 10 fractions, 24 Gy in 12 fractions, and 36 Gy in 18 fractions.
Initially 1:1 randomization to PCI:No PCI, then revised to 3:2 (PCI:No PCI)
Neurocognitive portion of trial was optional.
125 of 314 patients (40%) with baseline neurocognitive testing available.
59 of 314 patients (19%) with 6-month testing results available.
|
III
|
Results
No significant difference on multiple neurocognitive tests between PCI and No PCI at 6-months and 1-year. Cognitive impairment on study entry was seen on study entry in up to 42% of patients
Authors conclusion:
In both groups, there was similar degree of impairment of cognitive function and QOL before PCI. No difference in neurocognitive detriment between PCI and control in this patient population without brain metastases
Comments and conclusions:
Used simple proportions to compare cognitive decline at each time point. Designated as class III since patient numbers were relatively small at all time points, and neurocognitive testing was only available on 40% of patients at baseline.
|
|
Arriagada et al48 (1995)
|
Study description
Primary endpoint of multi-institutional French phase III RCT
SCLC histology
Included patients with SCLC, without brain metastases, with complete remission after induction therapy.
Treatment regimen
PCI
No PCI
PCI was 24 Gy in 8 fractions
Neuropsychologic assessments performed by neurologists, N = 294
|
II
|
Results
41% of all patients did not have neurocognitive abnormalities at baseline
Number of patients free from any abnormalities at baseline:
PCI: 50
No PCI: 44
2-year cumulative incidence of negative change in cognitive “higher functions” 36% (control) vs 30% (PCI), p = NS
PCI 30%
No PCI 36%, p = NS
Authors conclusion
Prophylactic cranial irradiation given to patients with small cell lung cancer in complete remission decreases the risk of brain metastasis threefold without a significant increase in complications. No difference in neurocognitive detriment between PCI and control in this patient population without brain metastases
Comments and conclusions
Used cumulative incidence for cognitive dysfunction endpoint. Designated as class II since “higher functions” were not defined, in addition to the lack of definition of criteria used to define decline.
|
BID, twice daily; COWAT, Controlled Oral Word Association Test; EORTC, European Organisation for Research and Treatment of Cancer; HRQOL, health-related quality of life; HVLT, Hopkins Verbal Learning Test; KPS, Karnofsky Performance Scale; MMSE, Mini-Mental State Examination; ND, neurocognitive decline; NS, not significant; PCI, prophylactic cranial irradiation; QOL, quality of life; RCT, randomized controlled trial; SCLC, small cell lung cancer; TMT, Trail Making Test.
Table 5. Neurocognitive outcomes of whole brain radiation therapy and local therapy versus local therapy only
|
Author and Year
|
Description of Study
|
Data Class
|
Conclusions
|
|
Soffietti et al51 (2013)
|
Study description
Secondary endpoint of European multi-institutional phase III RCT
Patient population
Patients with 1-3 brain metastases
Treatment regimen
Local only: local therapy alone with SRS or surgery (n = 179)
Local + WBRT (n = 180)
Local therapy either SRS (n = 199) or surgery (n = 160)
HRQOL measured with the EORTC Quality of Life Questionnaire C30 and the EORTC QLQ Brain Cancer Module
N = 341 with baseline HRQOL data
|
II
|
Results
EORTC QLQ C30 cognitive functioning score mean difference at 12 months
Local vs local + WBRT mean difference = -10.8 points (p < 0.05)
Mean EORTC QLQ C30 cognitive functioning score at 12 months
Local: 80.4
Local + WBRT: 69.7 (p = 0.05)
Authors conclusions
Adjuvant WBRT after surgery or SRS of a limited number of brain metastases may negatively impact some aspects of HRQOL, including self-reported cognitive functioning.
Comments and conclusions
Overall, patients treated with surgery or SRS only reported better HRQOL scores than did patients who also received WBRT. Most scores, which differed significantly during the first time points, had a tendency to recover. The positive effect of WBRT in decreasing the rate of intracranial progression and modestly improving the progression-free survival did not translate into an advantage in terms of HRQOL. Designated as class II since cognitive functioning was a secondary endpoint.
|
|
Chang et al50 (2009)
|
Study description
Primary endpoint of single institutional phase III RCT
Patient population
Patients with 1-3 brain metastases
Treatment regimen
SRS alone (n = 30)
SRS + WBRT (n = 28)
WBRT dose: 30 Gy in 12 fractions
Primary endpoint: significant deterioration of HTLV-R total recall at 4 months defined as ≥5 points drop from baseline.
Bayesian analysis
Trial enrollment stopped after 58 patients enrolled due to significant differences.
|
I
|
Results
HTLV-R significant deterioration rates at 4 months
Total recall:
SRS: 24%
SRS + WBRT: 52%
Delayed recall:
SRS: 6%
SRS + WBRT: 22%
Delayed recognition:
SRS: 0%
SRS + WBRT: 11%
Authors conclusions
Patients treated with SRS + WBRT were at a greater risk of a significant decline in learning and memory function by 4 months compared with the group that received SRS alone
Comments and conclusions
Significantly longer overall survival in patients treated with SRS alone as compared to SRS + WBRT. Given that this is a finding not found in other studies, thought to possibly be indicative of more favorable prognostic factors in SRS alone group.
|
|
Aoyama et al49 (2007)
|
Study description
Secondary endpoint of Japanese multi-institutional phase III RCT
Patients with 1-4 brain metastases
Treatment regimen
SRS alone (n = 67)
SRS + WBRT (n = 65)
WBRT dose: 30 Gy in 10 fractions
Neurocognition measured with MMSE.
110 of 132 randomized patients (83%) had baseline MMSE scores available.
|
II
|
Results
Average baseline MMSE did not differ significantly between treatment groups (p = 0.47).
Median MMSE score at 12 months
SRS alone: 28
SRS+WBRT: 27
Actuarial rate of MMSE preservation (decline < 3 points) at 12 months
SRS alone: 59.3%
SRS+WBRT: 76.1% (p = NS)
Actuarial rate of MMSE preservation (decline < 3 points) at 24 months
SRS alone: 51.9%
SRS+WBRT: 68.5% (p = NS)
Average duration until MMSE deterioration
SRS alone: 7.6 months
SRS+WBRT: 16.5 months (p = 0.05)
Authors conclusion
Intracranial control is the most important factor for stabilizing neurocognitive function. Addition of WBRT stabilized neurocognition in the intermediate term due to improved intracranial control, however WBRT may be associated with long-term adverse effects on neurocognition.
Comments and conclusions
Designated as class II since MMSE is a relatively insensitive measure of neurocognition and may miss more subtle changes.
|
Table 6. Effect of pharmacologic agents or whole brain radiation therapy techniques on neurocognitive decline
|
Author and Year
|
Description of Study
|
Data Class
|
Conclusions
|
|
Rapp et al53 (2015)
|
Study description
Primary endpoint of multi-institutional phase III RCT of donepezil versus placebo.
Patient eligibility
Patients with either primary or secondary brain tumors receiving partial brain (60%) or WBRT (40%) of at least 30 Gy ≥6 months before enrollment.
27% metastatic brain tumors
7% PCI
66% primary brain tumors
Treatment regimens
Donepezil: n = 99
Placebo: n = 99
Donepezil single daily 5-mg dose for 6 weeks, which was escalated to 10 mg per day for 18 weeks if well tolerated.
Primary endpoint: overall cognitive performance after 24 weeks of therapy
|
II
|
Results
24 week results:
Patients in both groups showed improved cognitive function at 24 weeks, but there was no difference in overall cognitive composite score between arms (p = 0.48)
No significant differences between groups except for memory recognition (p = 0.027), memory discrimination (p = 0.007), and motor speed and dexterity (p = 0.016)
The benefits of donepezil greater for those who were more cognitively impaired at baseline.
Author’s conclusions:
Treatment with donepezil did not significantly improve the overall composite score, but it did result in modest improvements in several cognitive functions, especially among patients with greater pretreatment impairments.
Comments and conclusions:
Assigned class II since only 40% of patients received WBRT. Donepezil only started 6 months after radiation therapy, providing a source of bias.
|
|
Brown et al56 (2013)
|
Study description
Primary endpoint of North American multi-institutional phase III RCT. Primary endpoint was decline in HVLT-R delayed recall at 24 weeks.
Patient eligibility
Patients with brain metastases (number not limited)
Treatment regimens
WBRT + memantine: n = 278
WBRT + placebo: n=276
WBRT 37.5 Gy in 15 fractions
Memantine dosing, starting before or during WBRT:
Week 1 5 mg qAM
Week 2 5 mg BID
Week 3 10mg qAM / 5 mg qPM
Week 4-24 10 mg BID
N = 473 with baseline scores available
Only 149 (53%) of 280 alive patients at 24 weeks had neurocognitive assessments and were analyzable.
|
I
|
Results
Median decline in HVLT-R delayed recall at 24 weeks
WBRT + memantine: 0
WBRT + placebo: -0.9 (p = 0.059, NS)
Probability of cognitive failure at 24 weeks:
WBRT + Memantine: 53.8%
WBRT + placebo: 64.9% (p = 0.01)
Authors’ conclusions
Memantine well tolerated. Although memantine was associated with less decline in the primary endpoint of delayed recall at 24 weeks, this lacked statistical significance possibly due to significant patient loss. Overall, patients treated with memantine had better cognitive function over time; specifically, memantine delayed time to cognitive decline and reduced the rate of decline in memory, executive function, and processing speed in patients receiving WBRT.
|
|
Gondi et al54 (2014)
|
Study description
Primary endpoint of multi-institutional North American phase II single arm trial of hippocampal avoidance (HA). Results compared with historical control of control arm of previous phase III RCT. Primary endpoint was decline in HVLT-R delayed recall (DR) at 4 months as compared with standard arm of PCI-P-120-9801 phase III trial using WBRT 30 Gy in 10 fractions without HA.
Patient eligibility
Patients with brain metastases outside a 5-mm margin around either hippocampus.
Treatment regiment
Patients treated with HA (n=113) during WBRT to 30 Gy in 10 fractions.
Hippocampal D100 goal <9 Gy and max point dose goal <16 Gy
100 patients with baseline scores available.
42 patients with scores analyzable at 4 months.
|
II
|
Results:
42 patients analyzable for primary endpoint at 4 months (71% of alive patients)
Mean relative HVLT-R DR decline between baseline and 4 months
HA: 7%
Historical controls: 30% (p = 0.0003)
Probability of HVLT-R total recall significant deterioration by 4 months
HA: 19%
Probability of HVLT-R DR significant deterioration by 4 months
HA: 33%
Intracranial progression within HA region
5% of patients with intracranial progression
3% of patients overall
Authors conclusions
Conformal avoidance of the hippocampus during WBRT is associated with preservation of memory and QOL as compared with historical series.
Comments and Conclusions
Designated as Class II since it was a Phase II study.
|
|
Butler et al55 (2007)
|
Study description
Secondary endpoint of multi-institutional phase III RCT of d-threo-methylphenidate HCl (d-MPH) versus placebo. Primary endpoint was fatigue subscale of the FACIT-F.
Patient eligibility
Patients with either primary brain tumors (n = 33) or brain metastases (n = 35) receiving partial brain RT or WBRT ≥25 Gy
Treatment regimens
RT + d-MPH: n = 34
RT + placebo: n = 34
d-MPH or placebo started by day 5 of RT.
Starting dose of d-MPH was 5 mg BID and was escalated by 5 mg BID to a maximum of 15 mg BID.
Study drug continued for 8 weeks post-RT.
QOL measured with FACT-Brain and FACIT-F and cognition measured with MMSE.
Trial closed after accrual of 68 of planned 162 patients due to slow accrual and withdrawal of financial support.
|
II
|
Results
Fatigue:
No difference in fatigue assessment at any time point up to 8 weeks post-RT between arms.
Baseline MMSE score
RT + d-MPH: 27.2
RT + placebo: 26.5 (p = NS)
MMSE 8 weeks post-RT
RT + d-MPH: 23.3
RT + placebo: 25.6 (p = NS)
Authors conclusions
Prophylactic use of d-MPH in brain tumor patients undergoing RT did not result in an improvement in QOL.
Comments and conclusions
Designated as Class II due to low patient accrual and reduced statistical power. Only a small number of patients receiving WBRT.
|
Table 7. Intracranial progression-free survival and overall survival following local therapy (surgery or stereotactic radiosurgery) alone or local therapy with whole brain radiation therapy
|
Author (Year)
|
Description of Study
|
Data Class
|
Conclusions
|
|
Kocher et al28 (2011)
|
Study description
RCT comparing WBRT to observation after SRS or surgical resection on duration of functional independence (WHO performance status)
Patient population
359 patients with 1-3 brain metastases with WHO performance status ≤2 who had previously undergone either surgical resection or SRS prior to randomized intervention
Treatment regimen
SRS + observation (n = 100)
SRS + WBRT (n = 99)
Surgery + observation (n = 79)
Surgery + WBRT (n = 81)
Local therapy + WBRT arm (180 total)
Local therapy + Observation (179 total)
WBRT 30 Gy in 10 fractions
|
II
|
Results
Survival with functional independence (time to WHO PS>2)
Observation: 10 months
WBRT: 9.5 months
(HR = 0.96, p = 0.71)
At 2 years, 22.3% and 22.6% were alive and independent in the observation and WBRT arms, respectively.
Progression-free survival
Observation: 3.4 months
WBRT: 4.6 months
(p = 0.020)
Overall survival
Observation: 10.9 months
WBRT: 10.7 months
(HR = 0.98, p = 0.89)
Author’s conclusions
After surgery or SRS, WBRT reduces the probability of intracranial relapses from 80% to 50%, and is most pronounced after surgery. This is translated into a modest PFS, but no improvement in OS. There was no difference in functional independence between the 2 groups.
Comments and conclusions
In well-performing patients with otherwise stable systemic disease and 1-3metastases, who are initially treated with either radiosurgery or surgery, WBRT can be withheld if serial imaging for follow-up is performed. Regarding the patients undergoing resection of a single lesion, because adjuvant irradiation substantially reduces the risk of recurrence in the tumor bed, postoperative local irradiation should be an option that is investigated. Designated class II since the primary endpoint was functional independence, not PFS or OS.
|
|
Aoyama et al59 (2006)
|
Study description
RCT comparing patients with 1-4 brain metastases receiving either WBRT + SRS or SRS alone on overall survival, recurrence, function, and cause of death. Study closed early due to poor accrual.
Patient population
132 patients with 1-4 brain metastases (each <3 cm in diameter).
No surgical resection performed prior to treatment.
Treatment regimen
WBRT + SRS (n = 65)
SRS alone (n = 67)
WBRT 30 Gy in 10 fractions
|
III
|
Results
Survival (median and 1-year actuarial survival rate)
WBRT + SRS: 7.5 months and 38.5%
SRS alone: 8.0 months and 28.4%
(p = 0.42)
Intracranial recurrence rate at 12 months
WBRT + SRS: 46.8%
SRS alone: 76.4%
(p < 0.001)
Salvage intracranial treatment
WBRT + SRS: 10 patients
SRS alone: 29 patients
(p < 0.001)
Cause of death: Neurological causes
WBRT + SRS: 22.8%
SRS alone: 19.3%
(p = 0.64)
Author’s conclusions
Compared to SRS alone, the use of WBRT + SRS did not improve survival for patients in this trial, but intracranial relapse occurred more frequently in those not receiving WBRT.
Comments and conclusions
Between both groups, there was no difference in OS, but higher rates of recurrence in the SRS only group lead to the more frequent need for salvage treatment. Assigned class III due to early closure of study due to poor accrual, resulting in lack of statistical power.
|
|
Patchell et al58 (1998)
|
Study description
RCT comparing patients with single brain metastases who underwent surgical resection followed by postoperative WBRT vs observation on tumor recurrence and survival.
Patient population
95 patients with single metastases to the brain treated with complete surgical resection
Treatment regimen
Surgery + WBRT (n = 49)
surgery + observation (n = 46)
WBRT 50.4 Gy in 28 fractions
Primary end point: intracranial recurrence
Secondary end points: Overall survival, cause of death, and preservation of ability to function independently
|
I
|
Results
Tumor recurrence
Surgery + WBRT: 18%
surgery + observation: 70%
(p < 0.001)
WBRT prevented recurrence at the site of original metastases (10% vs. 46%, p < 0.001) as well as other sites (14% vs. 37%, p < 0.01) vs. observation, respectively.
Death from neurological causes
Surgery + WBRT: 14%
surgery + observation: 44%
(p = 0.003)
Overall survival
Surgery + WBRT: 48 weeks
surgery + observation: 43 weeks
(p = 0.39)
Author’s conclusions
Postoperative WBRT after complete surgical resection of a single metastasis results in better control of disease in the brain and a reduction in the number of deaths due to neurological causes.Due to the decreased death due to neurologic causes, the authors recommended routine postoperative WBRT.
Comments and conclusions
Despite the reduction in brain recurrence rates and neurologic deaths, postoperative WBRT did not result in an increased survival or improvement in the length of time patients were able to function independently.
|
Gy, Gray; HR, hazard ratio; OS, overall survival; PFS, progression-free survival; SRS, stereotactic radiosurgery; WBRT, whole brain radiation therapy; WHO, World Health Organization.
REFERENCES
1. Gaspar LE, Mehta MP, Patchell RA, et al. The role of whole brain radiation therapy in the management of newly diagnosed brain metastases: a systematic review and evidence-based clinical practice guideline. J Neurooncol. Jan 2010;96(1):17-32.
2. Borgelt B, Gelber R, Larson M, Hendrickson F, Griffin T, Roth R. Ultra-rapid high dose irradiation schedules for the palliation of brain metastases: final results of the first two studies by the Radiation Therapy Oncology Group. Int J Radiat Oncol Biol Phys. Dec 1981;7(12):1633-1638.
3. Chatani M, Matayoshi Y, Masaki N, Inoue T. Radiation therapy for brain metastases from lung carcinoma. Prospective randomized trial according to the level of lactate dehydrogenase. Strahlenther Onkol. Mar 1994;170(3):155-161.
4. Chatani M, Teshima T, Hata K, Inoue T, Suzuki T. Whole brain irradiation for metastases from lung carcinoma. A clinical investigation. Acta Radiol Oncol. Jul-Aug 1985;24(4):311-314.
5. Davey P, Hoegler D, Ennis M, Smith J. A phase III study of accelerated versus conventional hypofractionated whole brain irradiation in patients of good performance status with brain metastases not suitable for surgical excision. Radiother Oncol. Aug 2008;88(2):173-176.
6. Haie-Meder C, Pellae-Cosset B, Laplanche A, et al. Results of a randomized clinical trial comparing two radiation schedules in the palliative treatment of brain metastases. Radiother Oncol. Feb 1993;26(2):111-116.
7. Komarnicky LT, Phillips TL, Martz K, Asbell S, Isaacson S, Urtasun R. A randomized phase III protocol for the evaluation of misonidazole combined with radiation in the treatment of patients with brain metastases (RTOG-7916). Int J Radiat Oncol Biol Phys. Jan 1991;20(1):53-58.
8. Kurtz JM, Gelber R, Brady LW, Carella RJ, Cooper JS. The palliation of brain metastases in a favorable patient population: a randomized clinical trial by the Radiation Therapy Oncology Group. Int J Radiat Oncol Biol Phys. Jul 1981;7(7):891-895.
9. Murray KJ, Scott C, Greenberg HM, et al. A randomized phase III study of accelerated hyperfractionation versus standard in patients with unresected brain metastases: a report of the Radiation Therapy Oncology Group (RTOG) 9104. Int J Radiat Oncol Biol Phys. Oct 1 1997;39(3):571-574.
10. Priestman TJ, Dunn J, Brada M, Rampling R, Baker PG. Final results of the Royal College of Radiologists' trial comparing two different radiotherapy schedules in the treatment of cerebral metastases. Clin Oncol (R Coll Radiol). 1996;8(5):308-315.
11. Sause WT, Scott C, Krisch R, et al. Phase I/II trial of accelerated fractionation in brain metastases RTOG 85-28. Int J Radiat Oncol Biol Phys. Jul 15 1993;26(4):653-657.
12. Bach F, Sorensen JB, Adrian L, et al. Brain relapses in chemotherapy-treated small cell lung cancer: a retrospective review of two time-dose regimens of therapeutic brain irradiation. Lung Cancer. Sep 1996;15(2):171-181.
13. Conill C, Jorcano S, Domingo-Domenech J, et al. Whole brain irradiation and temozolomide based chemotherapy in melanoma brain metastases. Clin Transl Oncol. Apr 2006;8(4):266-270.
14. Nieder C, Berberich W, Nestle U, Niewald M, Walter K, Schnabel K. Relation between local result and total dose of radiotherapy for brain metastases. Int J Radiat Oncol Biol Phys. Sep 30 1995;33(2):349-355.
15. Rades D, Schild SE, Lohynska R, Veninga T, Stalpers LJ, Dunst J. Two radiation regimens and prognostic factors for brain metastases in nonsmall cell lung cancer patients. Cancer. Sep 1 2007;110(5):1077-1082.
16. Rades D, Haatanen T, Schild SE, Dunst J. Dose escalation beyond 30 grays in 10 fractions for patients with multiple brain metastases. Cancer. Sep 15 2007;110(6):1345-1350.
17. Rades D, Bohlen G, Dunst J, et al. Comparison of short-course versus long-course whole-brain radiotherapy in the treatment of brain metastases. Strahlenther Onkol. Jan 2008;184(1):30-35.
18. Nieder C, Nestle U, Niewald M, Schnabel K. Accelerated radiotherapy for brain metastases. Radiother Oncol. Oct 1997;45(1):17-22.
19. Graham PH, Bucci J, Browne L. Randomized comparison of whole brain radiotherapy, 20 Gy in four daily fractions versus 40 Gy in 20 twice-daily fractions, for brain metastases. Int J Radiat Oncol Biol Phys. Jul 1 2010;77(3):648-654.
20. Saha A GS, Roy C, Kayal P. Treatment outcomes in patients with multiple brain
metastases: A prospective randomized study. . Clinical Cancer Investigation Journal. 2014;3(4):269-275.
21. Sayed M. Different fractionation in whole brain irradiation for multiple brain metastases
Middle East Journal of Cancer. 2015;6(2):85-90.
22. Fowler J. Practical time-dose evaluations, or how to stop worrying and learn to love linear quadratics. In: Purdy JA PC, Vijayakumar S, ed. Technical Basis of Radiation Therapy: Practical Clinical Applications . 4 ed: Springer Berlin Heidelberg; 2006:3-31.
23. Jones B, Dale RG, Deehan C, Hopkins KI, Morgan DA. The role of biologically effective dose (BED) in clinical oncology. Clin Oncol (R Coll Radiol). 2001;13(2):71-81.
24. Caravatta L, Deodato F, Ferro M, et al. Results of a phase II study of Short-course Accelerated Radiation Therapy (SHARON) for multiple brain metastases. Am J Clin Oncol. Aug 2015;38(4):395-400.
25. Rodrigues G, Yartsev S, Tay KY, Pond GR, Lagerwaard F, Bauman G. A phase II multi-institutional study assessing simultaneous in-field boost helical tomotherapy for 1-3 brain metastases. Radiation oncology (London, England). 2012;7:42.
26. Levegrun S, Pottgen C, Wittig A, Lubcke W, Abu Jawad J, Stuschke M. Helical tomotherapy for whole-brain irradiation with integrated boost to multiple brain metastases: evaluation of dose distribution characteristics and comparison with alternative techniques. Int J Radiat Oncol Biol Phys. Jul 15 2013;86(4):734-742.
27. Lee DS, Kim YS, Lee CG, et al. Early volumetric change and treatment outcome of metastatic brain tumors after external beam radiotherapy: differential radiotherapy for brain metastasis. Clin Transl Oncol. Nov 2013;15(11):889-896.
28. Kocher M, Soffietti R, Abacioglu U, et al. Adjuvant whole-brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: results of the EORTC 22952-26001 study. J Clin Oncol. Jan 10 2011;29(2):134-141.
29. Sundstrom JT, Minn H, Lertola KK, Nordman E. Prognosis of patients treated for intracranial metastases with whole-brain irradiation. Ann Med. Jun 1998;30(3):296-299.
30. Lee HL, Chung TS, Ting LL, et al. EGFR mutations are associated with favorable intracranial response and progression-free survival following brain irradiation in non-small cell lung cancer patients with brain metastases. Radiation oncology (London, England). Oct 30 2012;7:181.
31. Gow CH, Chien CR, Chang YL, et al. Radiotherapy in lung adenocarcinoma with brain metastases: effects of activating epidermal growth factor receptor mutations on clinical response. Clin Cancer Res. Jan 1 2008;14(1):162-168.
32. Wolstenholme V, Hawkins M, Ashley S, Tait D, Ross G. HER2 significance and treatment outcomes after radiotherapy for brain metastases in breast cancer patients. Breast. Dec 2008;17(6):661-665.
33. Borgelt B, Gelber R, Kramer S, et al. The palliation of brain metastases: final results of the first two studies by the Radiation Therapy Oncology Group. Int J Radiat Oncol Biol Phys. Jan 1980;6(1):1-9.
34. Sperduto PW, Kased N, Roberge D, et al. Effect of tumor subtype on survival and the graded prognostic assessment for patients with breast cancer and brain metastases. Int J Radiat Oncol Biol Phys. Apr 1 2012;82(5):2111-2117.
35. Andrews DW, Scott CB, Sperduto PW, et al. Whole brain radiation therapy with or without stereotactic radiosurgery boost for patients with one to three brain metastases: phase III results of the RTOG 9508 randomised trial. Lancet. May 22 2004;363(9422):1665-1672.
36. Solomon BJ, Cappuzzo F, Felip E, et al. Intracranial Efficacy of Crizotinib Versus Chemotherapy in Patients With Advanced ALK-Positive Non-Small-Cell Lung Cancer: Results From PROFILE 1014. J Clin Oncol. Aug 20 2016;34(24):2858-2865.
37. Schuler M, Wu YL, Hirsh V, et al. First-Line Afatinib versus Chemotherapy in Patients with Non-Small Cell Lung Cancer and Common Epidermal Growth Factor Receptor Gene Mutations and Brain Metastases. J Thorac Oncol. Mar 2016;11(3):380-390.
38. Iuchi T, Shingyoji M, Sakaida T, et al. Phase II trial of gefitinib alone without radiation therapy for Japanese patients with brain metastases from EGFR-mutant lung adenocarcinoma. Lung Cancer. Nov 2013;82(2):282-287.
39. Selek U, Chang EL, Hassenbusch SJ, 3rd, et al. Stereotactic radiosurgical treatment in 103 patients for 153 cerebral melanoma metastases. Int J Radiat Oncol Biol Phys. Jul 15 2004;59(4):1097-1106.
40. Liew DN, Kano H, Kondziolka D, et al. Outcome predictors of Gamma Knife surgery for melanoma brain metastases. Clinical article. Journal of neurosurgery. Mar 2011;114(3):769-779.
41. Mori Y, Kondziolka D, Flickinger JC, Kirkwood JM, Agarwala S, Lunsford LD. Stereotactic radiosurgery for cerebral metastatic melanoma: factors affecting local disease control and survival. Int J Radiat Oncol Biol Phys. Oct 1 1998;42(3):581-589.
42. Fogarty GB, Hong A, Dolven-Jacobsen K, et al. First interim analysis of a randomised trial of whole brain radiotherapy in melanoma brain metastases confirms high data quality. BMC Res Notes. May 08 2015;8:192.
43. Wolfson AH, Bae K, Komaki R, et al. Primary analysis of a phase II randomized trial Radiation Therapy Oncology Group (RTOG) 0212: impact of different total doses and schedules of prophylactic cranial irradiation on chronic neurotoxicity and quality of life for patients with limited-disease small-cell lung cancer. Int J Radiat Oncol Biol Phys. Sep 1 2011;81(1):77-84.
44. Le Pechoux C, Laplanche A, Faivre-Finn C, et al. Clinical neurological outcome and quality of life among patients with limited small-cell cancer treated with two different doses of prophylactic cranial irradiation in the intergroup phase III trial (PCI99-01, EORTC 22003-08004, RTOG 0212 and IFCT 99-01). Ann Oncol. May 2011;22(5):1154-1163.
45. Sun A, Bae K, Gore EM, et al. Phase III trial of prophylactic cranial irradiation compared with observation in patients with locally advanced non-small-cell lung cancer: neurocognitive and quality-of-life analysis. J Clin Oncol. Jan 20 2011;29(3):279-286.
46. Slotman BJ, Mauer ME, Bottomley A, et al. Prophylactic cranial irradiation in extensive disease small-cell lung cancer: short-term health-related quality of life and patient reported symptoms: results of an international Phase III randomized controlled trial by the EORTC Radiation Oncology and Lung Cancer Groups. J Clin Oncol. Jan 1 2009;27(1):78-84.
47. Gregor A, Cull A, Stephens RJ, et al. Prophylactic cranial irradiation is indicated following complete response to induction therapy in small cell lung cancer: results of a multicentre randomised trial. United Kingdom Coordinating Committee for Cancer Research (UKCCCR) and the European Organization for Research and Treatment of Cancer (EORTC). Eur J Cancer. Oct 1997;33(11):1752-1758.
48. Arriagada R, Le Chevalier T, Borie F, et al. Prophylactic cranial irradiation for patients with small-cell lung cancer in complete remission. J Natl Cancer Inst. Feb 1 1995;87(3):183-190.
49. Aoyama H, Tago M, Kato N, et al. Neurocognitive function of patients with brain metastasis who received either whole brain radiotherapy plus stereotactic radiosurgery or radiosurgery alone. Int J Radiat Oncol Biol Phys. Aug 1 2007;68(5):1388-1395.
50. Chang EL, Wefel JS, Hess KR, et al. Neurocognition in patients with brain metastases treated with radiosurgery or radiosurgery plus whole-brain irradiation: a randomised controlled trial. The Lancet. Oncology. Nov 2009;10(11):1037-1044.
51. Soffietti R, Kocher M, Abacioglu UM, et al. A European Organisation for Research and Treatment of Cancer phase III trial of adjuvant whole-brain radiotherapy versus observation in patients with one to three brain metastases from solid tumors after surgical resection or radiosurgery: quality-of-life results. J Clin Oncol. Jan 1 2013;31(1):65-72.
52. Brown PD, Jaeckle K, Ballman KV, et al. Effect of Radiosurgery Alone vs Radiosurgery With Whole Brain Radiation Therapy on Cognitive Function in Patients With 1 to 3 Brain Metastases: A Randomized Clinical Trial. Jama. Jul 26 2016;316(4):401-409.
53. Rapp SR, Case LD, Peiffer A, et al. Donepezil for Irradiated Brain Tumor Survivors: A Phase III Randomized Placebo-Controlled Clinical Trial. J Clin Oncol. May 20 2015;33(15):1653-1659.
54. Gondi V, Pugh SL, Tome WA, et al. Preservation of memory with conformal avoidance of the hippocampal neural stem-cell compartment during whole-brain radiotherapy for brain metastases (RTOG 0933): a phase II multi-institutional trial. J Clin Oncol. Dec 1 2014;32(34):3810-3816.
55. Butler JM, Jr., Case LD, Atkins J, et al. A phase III, double-blind, placebo-controlled prospective randomized clinical trial of d-threo-methylphenidate HCl in brain tumor patients receiving radiation therapy. Int J Radiat Oncol Biol Phys. Dec 1 2007;69(5):1496-1501.
56. Brown PD, Pugh S, Laack NN, et al. Memantine for the prevention of cognitive dysfunction in patients receiving whole-brain radiotherapy: a randomized, double-blind, placebo-controlled trial. Neuro Oncol. Oct 2013;15(10):1429-1437.
57. Auperin A, Arriagada R, Pignon JP, et al. Prophylactic cranial irradiation for patients with small-cell lung cancer in complete remission. Prophylactic Cranial Irradiation Overview Collaborative Group. N Engl J Med. Aug 12 1999;341(7):476-484.
58. Patchell RA, Tibbs PA, Regine WF, et al. Postoperative radiotherapy in the treatment of single metastases to the brain: a randomized trial. Jama. Nov 04 1998;280(17):1485-1489.
59. Aoyama H, Shirato H, Tago M, et al. Stereotactic radiosurgery plus whole-brain radiation therapy vs stereotactic radiosurgery alone for treatment of brain metastases: a randomized controlled trial. Jama. Jun 7 2006;295(21):2483-2491.
60. Sahgal A, Aoyama H, Kocher M, et al. Phase 3 trials of stereotactic radiosurgery with or without whole-brain radiation therapy for 1 to 4 brain metastases: individual patient data meta-analysis. Int J Radiat Oncol Biol Phys. Mar 15 2015;91(4):710-717.
61. Rades D, Hornung D, Veninga T, Schild SE, Gliemroth J. Single brain metastasis: radiosurgery alone compared with radiosurgery plus up-front whole-brain radiotherapy. Cancer. Jun 1 2012;118(11):2980-2985.
62. McPherson CM, Suki D, Feiz-Erfan I, et al. Adjuvant whole-brain radiation therapy after surgical resection of single brain metastases. Neuro Oncol. Jul 2010;12(7):711-719.
63. Yamamoto M, Serizawa T, Shuto T, et al. Stereotactic radiosurgery for patients with multiple brain metastases (JLGK0901): a multi-institutional prospective observational study. Lancet Oncol. Apr 2014;15(4):387-395.
64. Chang WS, Kim HY, Chang JW, Park YG, Chang JH. Analysis of radiosurgical results in patients with brain metastases according to the number of brain lesions: is stereotactic radiosurgery effective for multiple brain metastases? Journal of neurosurgery. Dec 2010;113 Suppl:73-78.
65. Bhatnagar AK, Flickinger JC, Kondziolka D, Lunsford LD. Stereotactic radiosurgery for four or more intracranial metastases. Int J Radiat Oncol Biol Phys. Mar 1 2006;64(3):898-903.
66. Gaspar L, Scott C, Rotman M, et al. Recursive partitioning analysis (RPA) of prognostic factors in three Radiation Therapy Oncology Group (RTOG) brain metastases trials. Int J Radiat Oncol Biol Phys. Mar 1 1997;37(4):745-751.
67. Banfill KE, Bownes PJ, St Clair SE, Loughrey C, Hatfield P. Stereotactic radiosurgery for the treatment of brain metastases: impact of cerebral disease burden on survival. British journal of neurosurgery. Oct 2012;26(5):674-678.
68. Mulvenna P, Nankivell M, Barton R, et al. Dexamethasone and supportive care with or without whole brain radiotherapy in treating patients with non-small cell lung cancer with brain metastases unsuitable for resection or stereotactic radiotherapy (QUARTZ): results from a phase 3, non-inferiority, randomised trial. Lancet. Oct 22 2016;388(10055):2004-2014.
Source: Neurosurgery